Belize Aquaculture identifies challenges hindering shrimp culture for the next century
Conventional wisdom for farming of (Litopenaeus vannamei) in the Americas involves semi-intensive culture in large ponds, routine water exchange, and reliance on locally available wild seed stock or broodstock. Because water exchange is the most important tool in management of these ponds, preferred sites are thought to be those located beside the source of water. Relatively low land elevations are usually selected in order to use low head, high volume pumps. Only soils with a high clay content and low acidity would be considered acceptable for the development of most shrimp projects. Such were the paradigms of shrimp farming in Belize in 1985, when Barry Bowen, a land owner and businessman in Belize, started looking into shrimp farming as a way of diversifying the agronomics of the country.
At that time, Belize did not have a wild source of cultivable shrimp, and hatchery/maturation technology seemed uncertain, so Barry put his plans on hold. Then, in the 1990’s he again considered the feasibility of shrimp farming in Belize, and again hesitated when he saw the impact that Taura had on existing shrimp farms in Belize. Also at that time, environmental groups started raising issues about the sustainability of shrimp farming, which led to concern about disruption of shrimp markets in the future. It seemed clear that alternative technologies were needed in Belize to culture non-indigenous shrimp in an environmentally friendly way with high land use efficiency on property outside of the hurricane zone.
A trip to South Carolina and the Waddell Mariculture Center in the summer of 1995 increased Barry’s awareness of alternative types of shrimp culture technologies. In 1996, when shrimp farms throughout Belize simultaneously dried out and succeeded in eliminating the Taura virus, the timing seemed right to test these new concepts. This was the background that led to the development of Belize Aquaculture, Ltd. – a testing ground for new paradigms of shrimp farming. Two years of exciting data have now been collected, and we are happy to share these in the spirit of improving the sustainability of the industry. The editor of the Advocate has requested that we present a series of articles beginning with this general description of the farm and followed by more specific articles about our experiences with shrimp breeding, nutrition, pond management, health management and new technologies.
Objectives
In order to meet the basic objectives of being sustainable for many years, being environmentally friendly, and making use of land outside the hurricane zone with high efficiency; the project had to evaluate new technologies and combine existing technologies in unique ways. The entire project was based on ideas that were contrary to conventional shrimp farming wisdom:
- utilize a zero water exchange strategy
- target 11,400 kilograms per hectare of shrimp per cycle with 2.4 harvests per year
- locate ponds several miles from the coast in a pine ridge
- use turbine pumps to procure pure Caribbean sea water
- build the ponds in sandy, acidic soils
- produce postlarvae on site from broodstock imported from a certified SPF facility.
Since the concepts for Belize Aquaculture, Ltd. were quite unorthodox, the first phase was designed as a research and development effort to test the concepts before expanding. Construction of the R&D facility began on May 1996, and the first ponds were stocked in September 1997.
Our R&D phase established four goals:
- Produce 2.4 crops of 11,200 kilograms per hectare per crop for a period of two years, using a culture system based on zero water exchange, water re-use after harvest, HDPE lined ponds for rapid turn around between crops, construction of ponds in acidic sandy soils, and the exclusive use of SPF shrimp for reducing the possibility of viral introductions.
- Determine which size pond would be most appropriate for commercial application, and the engineering requirements to construct and operate such ponds.
- Determine which shrimp species and strains were most suited for this zero exchange, intensive culture system.
- Develop the operational skills and experience needed to successfully run such a system.
Components of the facility
The facility is located at Blair Atholl estate in the Stann Creek District of Belize, just West of the Placencia Lagoon. The estate has approximately 3,500 has of land, which is mostly pine savanna. Soils are deposits of acidic, sandy clay, and are low in nutrients and high in aluminum. Most of the land is at an elevation greater than 20 feet above sea level. The farm facility consists of:
- Sixteen 0.065 ha ponds, eight 0.37 ha ponds, one 1.0 ha pond and one 1.6 ha pond
- Two 0.7 ha settling basins
- Two 0.5 ha seawater reservoirs
- A maturation/hatchery complex
- A warehouse complex
- Four 230 kWh electric generators
- A housing complex for management and workers
- An acclimation station
- A brackish water creek pump station
- A seaside pump station.
The hatchery complex is designed to produce 2.5 million postlarvae (PL) per month, with the maturation units designed to produce 240 million nauplii per month.
Water supply system
Water management on the R&D facility was designed for flexibility. Water can be exchanged up to 15 percent per day if desired, and salinity can be controlled by pumping creek water. A seawater pump station has been set up on Placencia Peninsula. Using turbine pumps, seawater is pumped at a rate of 1,200 gallons per minute from 150 meters off the coast, to the main farm sea reservoir, a 3-km distance on the West side of Placencia lagoon. The seawater reservoir feeds seawater to both the hatchery and pond areas. Seawater can either be pumped directly to the ponds or can be gravity fed to the settling basins, where the water can be pumped to the ponds after preliminary fertilization or aging.
During the dry season salinity rises, and creek water can be pumped to the settling basins to reduce and adjust salinities. All water that is pumped from either the creek or from the sea is passed through a 250-micron filter bag. All drainage canals lead to the settling basins, so that when ponds are drained at harvest, the water can be retained, settled, and pumped back to the ponds. Water is recycled but not recirculated between ponds.
Pond design
Ponds are square-shaped, and have a shallow end of 1.4 meters sloping to a depth of 2.3 meters at the drain end. The 1.0 and 1.6 ha ponds are sloped to a center drain. All ponds are lined with either 30 or 40 mil HDPE plastic. Ponds have a stand pipe for water level control, which are connected to the drainpipes by a flange (which is removed for harvesting). Aeration varies with size of pond; the 0.065 ha pond has approximately 60 applied horsepower per hectare, the 0.37 ha pond has 28 applied horsepower per hectare, and the 1.0 and 1.6 ha ponds have 30 applied horsepower per hectare.
Aeration is provided by a combination of paddlewheel aerators placed in the corners of the ponds, and aspirator-type aerators placed in the center of the ponds. The aspirator-type devices are used to keep sediments and flock in suspension, which would otherwise settle in the center areas of the pond. Erosion is reduced by the use of erosion matting, and subsequent planting with St. Augustine grass.
Zero water exchange concept

Middle: Large amounts of foam on the surface of a pond four weeks after stocking.
Bottom: Bacterial floc from pond water readily sediments to the bottom of these one liter Imhoff cones.
The basis of the culture system in ponds is the promotion of a bacteria-dominated, stable ecological system, instead of the phytoplankton-dominated system, which can be highly unstable. The ponds are fed a combination organic mix and shrimp feed from the start of the cycle, at a rate which is much greater than would be consumed by the shrimp.
The idea is to promote the growth of bacteria, and establish a large outdoor bioreactor, or a system similar to a sewage oxidation pond. The ponds start out with a phytoplankton bloom, but by week 8-10 of culture, these blooms are replaced by bacterial flocs (Fig.1). Ponds can be easily differentiated as to the their stage of development. Young surface. Older ponds will turn brownish/blackish in color, and be free of foam on the water surface. Water from the older ponds is dominated by large organic/bacterial flocs that rapidly settle out if water agitation is removed.
Once the pond reaches this older stage they are highly stable, and can assimilate large amounts of organic inputs. Ammonia levels are generally under 2 ppm, pH generally ranges from 7.0 to 7.5, and dissolved oxygen levels range from 4.0 to 6.0 milligrams per liter. Feeding levels as high as 450 horsepower per hectare per day have been used.
Regarding the flocs, it is also very likely that these aid in the recycling of nutrients within the pond and shrimp population. Nitrogen assimilation efficiency is the percent nitrogen retained within the shrimp biomass, relative to the amount of nitrogen put into the pond either through feeds or inorganic fertilizers.
The implementation of most shrimp farming technologies result in nitrogen assimilation efficiencies of 20 to 25 percent. Belize Aquaculture has demonstrated that nitrogen assimilation efficiencies of greater than 45 percent are possible. This implies that nitrogen is being cycled several times through the shrimp, thereby significantly increasing the amount of nitrogen being retained by the shrimp biomass and reducing nitrogen waste.
Breeding
Other key factors to the strategy employed by Belize Aquaculture are the use of defined genetic strains of shrimp which are specific pathogen free, and the development of feeds with lower protein levels that do not create an overload of nitrogen and phosphorous in the pond, and yet promote the recycling of nutrients within the pond.
To date, three genetically distinct strains of L. vannamei have been cultured on the farm, and results indicate significant differences in performance among the three. In order to establish consistency, defined genetic strains that perform well need to be identified and further developed through selection processes.
Nutrition
The higher the protein and nutrient density in the feed, the more difficult it is for the zero exchange system to assimilate the residues. Feeds with protein levels higher than 30 percent have created more problems in the ponds and less consistent results than feeds with protein levels under 25 percent. Survival has been consistently higher when ponds were fed diets lower in protein. Because L. vannamei grows well on diets with protein levels less than 24 percent, the species is very well suited for culture in the zero water exchange systems.
Yields
The farm has just completed its second year of operations, and the results to date have surpassed the established goals. During the initial 23 months of operations, 193,180 kg of shrimp were harvested from 17.2 has of pond area (63 harvests). This represents an average yield of 11,231 kilograms per hectare per crop. The farm results are summarized in Table 1. During this period, 55 percent of all harvests produced yields greater than 13,600 kilograms per hectare. Harvests that have been lower than the 13,600 kilograms per hectare level were generally from ponds stocked with experimental Taura-resistant strains of L. vannamei or with L. stylirostris. The highest yield has been 27,200 kilograms per hectare, obtained in replicated 0.065 ha ponds combining 60 applied horsepower per hectare with AquaMats™ placed in the pond at a density of 150 per hectare (5 × 0.9 meters).

Pond performance and yield differences can be attributed to the following factors: stocking density, species/strain of shrimp being stocked, the time of year the pond was stocked, and the amount of aeration applied to the pond.
Higher stocking rates have resulted in proportionately higher yields, up to the point where the rate of aeration or the ability of the pond to assimilate inputs become limiting. Stocking rates greater than 120 per square meter generally resulted in yields greater than 13,600 kilograms per hectare when L. vannamei of the Mexican strain were stocked. Production seems to be increased by adding aeration up to 30 horsepower per hectare.
After this level of aeration is applied, production has not been increased efficiently by the addition of more aeration, as there seem to be other rate limiting factors effecting production. The introduction of AquaMats™ seems to have removed such a barrier, and thus the increase of yields in the small 0.0065 ha ponds from 18,200 kilograms per hectare (300 kilograms per horsepower aeration) to 27,200 kilograms per hectare (450 kilograms per hectare).
One of the biggest constraints to this new technology would be if large ponds could not be operated as efficiently as smaller ponds. The 1.6 ha pond, when stocked with L. vannamei (Mexican strain) at 118 per square meter produced 21,500 kg of shrimp. This is equivalent to 13,400 kilograms per hectare. The biggest challenge is not in growing the shrimp in larger ponds, but in the harvesting of such a large amount of shrimp over a very short period of time (Fig. 2).
Environmental sustainability
Certain interest groups have voiced many concerns about the possible effect shrimp farming activities have on the environment. Some of these concerns have included:
- Detrimental effects of nutrient loading on adjacent estuarine and oceanic waters.
- Destruction of mangrove and estuarine habitats.
- Effects of erosion on local habitats.
- Contamination of fresh ground waters with salts.
- Introduction of viral pathogens into local populations of crustaceans.
- Use of large amounts of fish meal to produce a smaller amount of shrimp; and
- The perturbation of large amounts of land to produce a relatively small amount of shrimp.
Belize Aquaculture is demonstrating that many of these concerns can be eliminated by adapting and developing alternative technologies for growing shrimp.
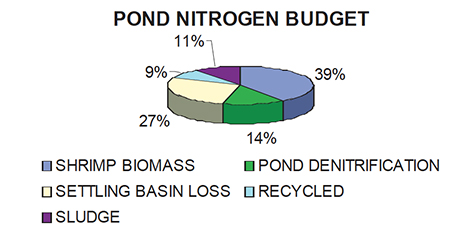
Zero exchange systems return very little nutrients back to the source estuarine and oceanic waters. All waste or exchanged water is directed through a settling basin, where the water is retained for seven days. During this period of settling, 60-70 percent of total nitrogen and 70-80 percent of total phosphorous are removed from the water. A nitrogen budget typical for a culture cycle that used 22 percent protein feeds, and retained the harvest water in the settling basin for seven days is presented in Fig. 3. For every 100 kg of nitrogen introduced into the culture system, 9 kg of nitrogen would be returned to the pond for the next culture cycle. Sludge that accumulates in the settling basin can be periodically removed, and used as a soil amendment after salts are leached from the material. Analysis has shown the sludge in the basin to have an organic content of over 65 percent.
The shrimp farm is constructed at above 20 feet elevation, away from any mangrove or estuarine areas. And each pond is lined with HDPE on the inside and erosion matting on the outside, preventing both seepage of salts into the freshwater aquifers and erosion of berm materials from the pond banks.
Finally, farm trials have shown that with proper feed formulation diets, with less than 10 percent fishmeal and 22 percent protein, can produce yields greater than 14,500 kilograms per hectare with growth rates exceeding 1.0 gram per week. This strategy results in one kilogram of shrimp being produced for every 0.6 kg of menhaden used in shrimp feeds.
Economics

Finally, Belize Aquaculture will not be successful unless it produces shrimp profitably. Costs have been similar to incurred costs using semi-intensive culture technologies. The farm average FCR is under 2.0 using a 24 percent protein diet (Fig. 4). The cost of seed is equal to that of any farm that produces shrimp with a 70 percent survival rate, and lower than for those farms which have survival rates below 70 percent. The labor cost/unit of production is lower, and the energy cost/unit of production is only slightly higher than semi-intensive culture. With increases in both generation and production efficiencies, the cost of energy will eventually be less than that of conventional technologies.
Conclusions
Shrimp farming is being challenged by both the rapid dissemination of viral diseases around the world and the perception that the industry is causing ill effects on the environment. Belize Aquaculture has tried to identify the challenges that would effect the business of growing shrimp for the next century. Through the adoption and further development of new technologies, the company is striving to establish a farming concept that will lead to the sustainable and profitable culture of shrimp well beyond the 21st century. Results to date have proven positive, and the project will continue the refinements and further adoptions that will be required to maintain success.
(Editor’s Note: This article was originally published in the August/October 1999 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-

Robins P. McIntosh
Belize Aquaculture, Ltd.
#1 King Street, P.O. Box 37
Belize City, Belize, Central America[116,101,110,46,108,116,98,64,97,117,113,97,101,122,105,108,101,98]
Related Posts

Intelligence
Changing paradigms in shrimp farming, part 2
The focus at Belize Aquaculture Ltd. was to use clean animals that perform well at high densities and in waters with high nutrient loads.

Intelligence
Changing paradigms in shrimp farming, part 3
Square-shaped and deep ponds may explain partially why Belize Aquaculture is able to produce a large amount of shrimp on a unit area basis.

Intelligence
Changing paradigms in shrimp farming, part 4
In high-density systems with zero water exchange, pond ecology shifts from an autotrophic, phytoplankton-based microbial community to a heterotrophic, bacterial-based community.

Intelligence
Changing paradigms in shrimp farming, part 5
A zero-water-exchange strategy to produce shrimp reduces the effluents and sediments that would normally be released to the environment by a typical intensive shrimp farm.

