Evaluations on L. vannamei conducted at Texas A&M University

An aquaculture microbial probiotic is a bacterial supplement (single or mixed culture of selected bacteria) added to a production system to modify or manipulate the microbial communities in the water and sediment, to reduce or eliminate selected pathogenic microorganisms, and to improve growth and survival of the cultured species. Manufacturers claim probiotics also improve water quality and lower organic sludge levels.
Probiotics have been reported effective as an inoculation to newly disinfected water in larval culture systems, where they reduce the need for antibiotics. The efficacy of probiotic additions to natural waters in grow-out systems is more controversial.
Testing protocol
With assistance from Tzachi Samocha and co-workers at Texas A&M University, we evaluated the routine use of a commercial bacterial supplement that claimed to improve water and sludge quality, and production of two shrimp species (Penaeus setiferus and Litopenaeus vannamei). Tests lasted three months, and were conducted in outdoor tanks under “zero water discharge” using a high aeration rate, stocking densities of 40 to 50 shrimp per square months, and both high- (45 percent) and low- (20 percent) protein diets.
We tested a commercial mixture of several species of the genus Bacillus (B. subtilis, B. megaterium, B. polymyxa, and B. licheniformis). All were non-pathogenic, viable, and naturally occurring bacteria that had not been genetically altered or engineered. Prebrewed microbial preparations were added to test tanks five times a week, per manufacturer specifications.
We monitored water quality, including dissolved oxygen, temperature, salinity, pH, Secchi disc, total ammonia-N, nitrite-N, nitrate-N, total phosphorus, reactive phosphorus, fiveday carbonaceous biochemical oxygen demand (cBOD5), chemical oxygen demand (COD), total suspended solids (TSS) and volatile suspended solids (VSS). At harvest, sediment slurry from each tank was sampled and analyzed for its relative dry weight, sediment volume, COD, cBOD5 and VSS content, and microbial respiration. Shrimp survival, mean final weight, final yield, and FCR were also determined.
Results
The studies revealed no significant differences between tanks treated with the commercial bacterial supplement and those that were not, for shrimp survival, final yield, mean final weight, and FCR with both diets for either shrimp species. The bacterial treatment did not produce any significant improvement on ammonia or nitrite removal, nitrate accumulation in the tanks, or any other water quality parameters monitored.
There were no significant differences in sludge parameters between the treated and untreated shrimp tanks. Therefore, addition of the bacterial supplement did not produce any measurable improvements in water or sediment quality, or shrimp yield over the untreated tanks.
Tests with other species
Claude Boyd and co-workers at the International Center for Aquaculture and Aquatic Environments at Auburn University in Alabama, USA, tested the same probiotic product in channel catfish ponds. They reported no significant differences in water quality parameters between treated and untreated ponds.
There was a higher survival rate in the probiotic-treated ponds, which ultimately led to greater fish production. However, fish had larger average weight in the untreated ponds.
Suhendra and co-workers reported that addition of this probiotic supplement to intensive tiger prawn (Penaeus monodon) cultures improved the cleanliness of water and sediment. But it is not possible to isolate the direct effect of the bacterial supplement from the possible contribution of all other factors that were modified, because different management strategies were followed in shrimp farms that participated in this study.
Conclusion
The benefits of using bacterial supplements to improve grow-out systems are still controversial. These supplements should be applied in ways that favor their establishment and activity, including direct introduction (e.g., by incorporating them in the feed), added surface area for microbial adhesion, manipulating the microbial population genetically by selection and enrichment, and by identifying and supplying factors that might otherwise limit beneficial microbial proliferation.
(Editor’s Note: This article was originally published in the October 2000 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Ami Horowitz, Ph.D.
UPAH Tech, Inc.
Shaker Heights, Ohio USA[109,111,99,46,111,111,104,97,121,64,104,99,101,116,104,97,112,117]
-
Sarah Horowitz, Ph.D.
UPAH Tech, Inc.
Shaker Heights, Ohio USA[109,111,99,46,111,111,104,97,121,64,104,99,101,116,104,97,112,117]
Tagged With
Related Posts

Health & Welfare
Biosecurity basics for shrimp aquaculture
Biosecurity in shrimp aquaculture is achieved by preventing the presence, growth, and spread of pathogenic microorganisms.
Responsibility
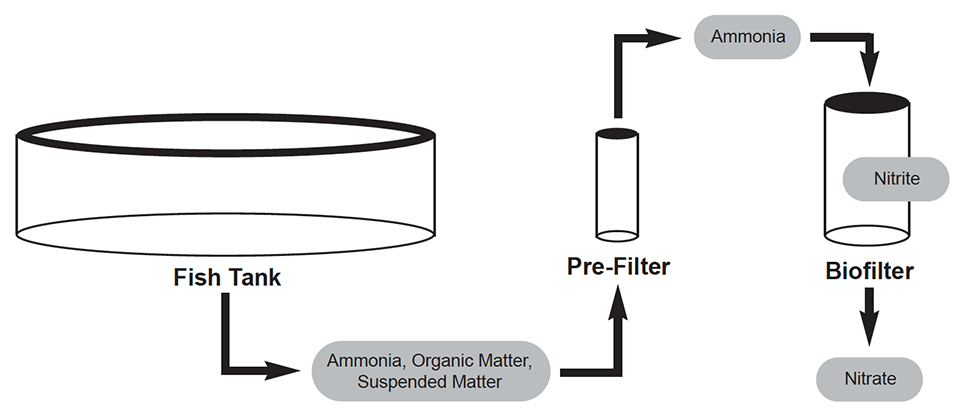
Improving biofiltration in recirculating aquaculture systems
The design and operation of biofilters is multidisciplinary, involving mechanical engineering, microbial ecology and aquaculture husbandry.
Responsibility
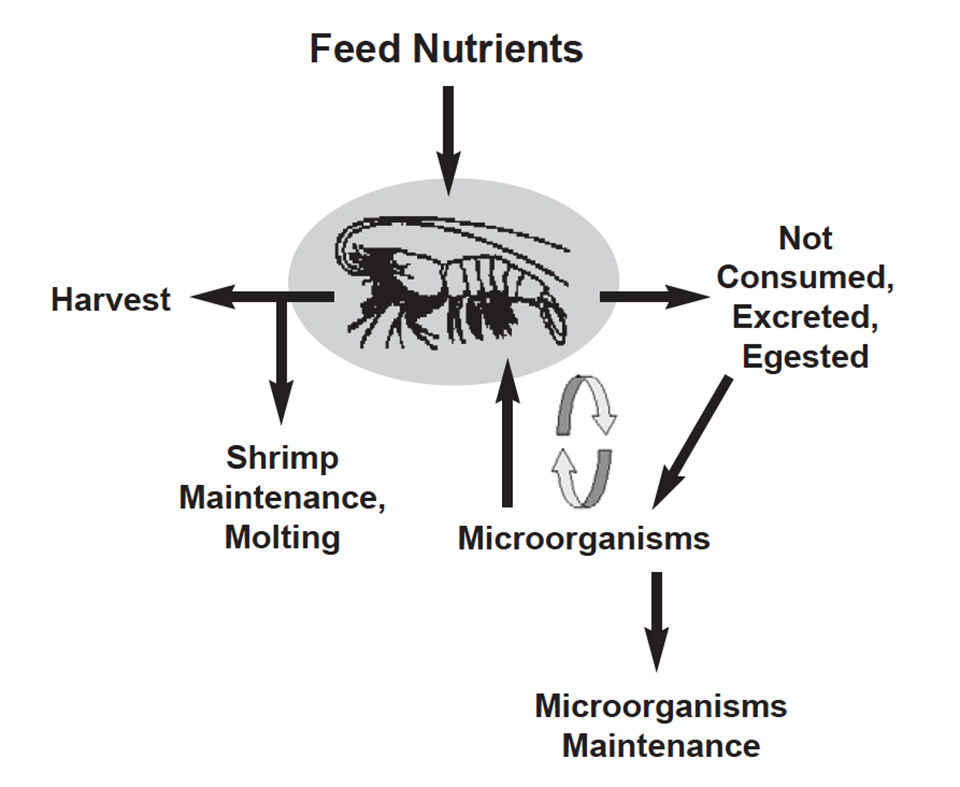
Microorganisms and feed management in aquaculture
Shrimp are the most commercially important aquaculture animals that utilize a significant amount of microorganisms in their diet.

Responsibility
Microbial testing identifies potential toxicity in polymer liners, biofilter materials
Although widely used in aquaculture applications, some synthetic polymer liners can release toxic substances into culture water and affect animal health.


