Rates of chemical processes increase in response to greater water temperature

Water temperature is a key water quality variable because it influences all other water quality variables and aquatic organisms, as well. Air temperature is controlled mainly by solar radiation, and it varies more or less predictably with season and time of day. Water in ponds and other outdoor aquaculture systems also receive direct insolation.
However, water warms more slowly than air because of its great specific heat of 1 calorie/gram/degrees-C. On sunny or partly cloudy days, pond water has its lowest temperature in the early morning and its highest temperature in the early afternoon – as does the air.
Water can store considerable heat because of its high specific heat. Heat stored in pond water is lost to the air when the air is cooler than the water. Because water releases heat rather slowly, brief periods of unseasonably cool weather do not cause drastic changes in water temperature in ponds. Nevertheless, if cool conditions persist for a few days, pond water temperature will equilibrate with the air temperature. When considered over periods longer than several days, water temperature closely tracks air temperature.
Water temperature influences the length of the growing season and the species that can be grown at a particular location. For example, tilapia, a tropical species, will die during winter in the temperate zone. Channel catfish, a warmwater, temperate zone species, can survive year-round in the temperate zone, but will not feed and grow during winter. Coldwater species do not survive well at temperatures above about 20 degrees-C, and warmwater species do not occur in cold climates.
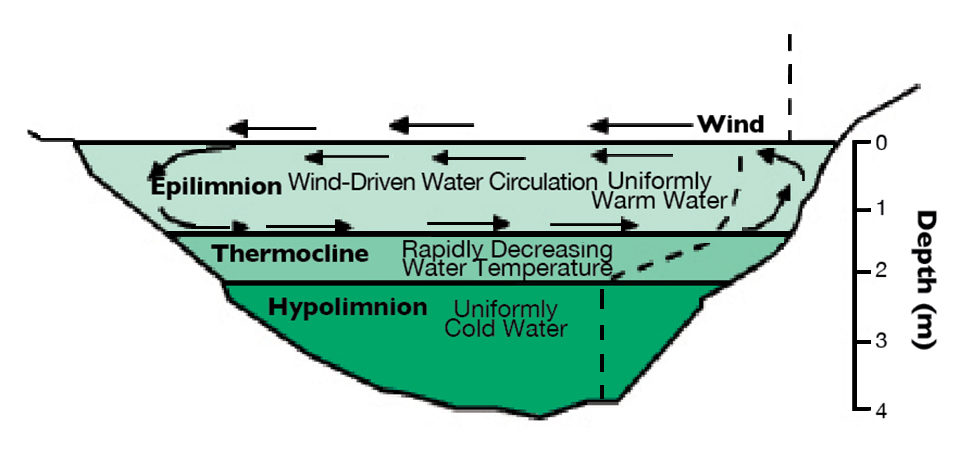
Thermal stratification
Light adsorption and heating of water are magnified by the presence of suspended particles, especially plankton and other organic matter. Light penetration into water declines exponentially with depth, so the surface layer heats much faster than deeper layers.
Warm water is less dense than cooler water and floats on top of it in the absence of strong mixing forces. In lakes and even small bodies of water greater than 1.5 or 2.0 m in depth, the warmer surface layer can increase enough in density that winds are not strong enough to mix it with deeper, cooler and denser water during the warmer parts of the year. This phenomenon results in thermal stratification.
When air temperatures decline in the cooler seasons, surface water cools until its density is low enough that wind can mix it with deeper water. Thermal destratification also can occur in the summer in response to unseasonable cool spells, heavy rains or strong winds.
Thermal stratification obviously can occur in tropical waters. Small tropical water bodies typically stratify and destratify annually. Destratification may not be caused by falling air temperature, but as a result of stronger winds or heavy rains during the wet season. Rain water is cooler and denser than warm pond water and sinks, causing upwelling and destratification. Large, deep tropical lakes stratify but tend not to destratify annually. Most destratify on an irregular basis as a result of weather conditions.
The surface layer of a stratified water body is called the epilimnion. The deeper, cooler layer is known as the hypolimnion, and the transitional layer of rapidly changing water temperature between the two is known as the thermocline.
Water quality deterioration
Because the hypolimnion does not receive enough light for photosynthesis, and organic particles settle into it, its water quality often becomes impaired by dissolved oxygen depletion and high concentrations of organic matter and other reduced substances during stratification. Sudden destratification mixes epilimnetic water with hypolimnetic water, which can result in water quality deterioration throughout the water column and mortality of culture animals.
Destratification of lakes containing cage culture operations can be especially disastrous. Low dissolved-oxygen concentrations following sudden destratification have caused complete mortality of fish in cages. The likelihood of sudden destratification and its possible consequences on culture should be carefully assessed in selecting sites for cage farms.
Aeration mixing
Aquaculture ponds should not be deeper than 2.0 m to minimize the probability of thermal stratification. Shallow ponds may stratify during the day, but destratification will break up at night. Ponds also should be oriented with their long axes parallel to the prevailing winds to encourage wind mixing.
Mechanical aeration mixes ponds, but surface aeration may not prevent stratification of deep ponds. Two mechanical methods are available for avoiding destratification in small, deep water bodies. Vertical, axial-flow devices can be used to propel surface water downward to blend with deeper water and prevent layers of different densities from developing. Air diffusers placed in deep water induce upwelling and also prevent stratification.
Seasonality
Temperature has a pronounced effect on chemical reaction rates, the ability of water to hold dissolved oxygen and other gases, and growth and other physiological processes. According to Van Hoff’s law, a 10 degrees C increase in temperature will roughly double the rate of chemical reactions, including physiological processes and growth. The actual factor of increase in a process caused by a 10 degrees C temperature is called the Q10. To illustrate, if fish grow 1.8 times faster at 30 degrees than at 20 degrees-C, Q10 is 1.8.
Water temperatures at most locations vary between seasons, even in the tropics. This phenomenon is illustrated in Fig. 1 with data from Auburn, Alabama, USA, at 32 degrees 36’ N latitude and for Guayaquil, Ecuador, at 2 degrees 9’ S latitude.
A small difference in temperature can appreciably affect shrimp growth rate. Suppose shrimp grow at 1.20 gram per week during a period when water temperature averages 27 degrees-C. Assuming Q10 = 2 for shrimp growth, an increase in temperature to 29 degrees-C should increase growth rate to 1.44 grams per week. Of course, plants and animals have a range of temperature tolerance, and Van Hoff’s law does not apply outside the temperature range for optimum growth. In the example above, if 29 degrees-C is above the optimum temperature for shrimp growth, there would be a decrease in growth at the higher temperature. A study in Poland revealed that a difference in 1 degree-C from the mean seasonal temperature can alter common carp production in intensive systems by 1,000 kg/ha.
Animal health, reproduction
Water temperature has an indirect role in the health of aquatic animals because it influences the occurrence and outcome of infectious disease. The immune systems of aquatic animals generally function most efficiently within the temperature range for optimum growth. At higher or lower temperatures, the immune systems are less effective in preventing disease.
Rapid temperature changes also impair immune function. Each pathogen has an optimal temperature range. For example, the bacterium that causes enteric septicemia in channel catfish is most virulent at water temperatures of 22 to 28 degrees-C. This temperature range in the major catfish-farming area of the United States occurs in spring and autumn, so these are the times that major outbreaks of the disease occur.
Water temperature plays an important role in determining the rate of ovulation and milt production in fish under natural and induced situations. Timing of this physiological response is related to a degree-hour response – water temperature multiplied by the number of hours from the onset of ovary maturation or dosing when inducing ovulation until ovulation. Ovulation in common carp, for example, requires 240 to 290 degree-hours.
Water quality
Water temperature greatly influences water quality because the growth and metabolism of phytoplankton, bacteria and other microorganisms increase with increasing temperature. Moreover, water holds less oxygen at higher temperatures. Dissolved-oxygen depletion is much more likely to occur during hot weather than during cooler periods.
Rates of chemical processes such as ionization, mineral dissolution, adsorption and ion exchange also increase in response to greater water temperature. The onset of potentially harmful water quality events is faster in warmer weather. Aquaculture managers should be especially vigilant regarding water quality during the warmest months and periods of unusually high temperature.
(Editor’s Note: This article was originally published in the May/June 2010 print edition of the Global Aquaculture Advocate.)
Author
-

Claude E. Boyd, Ph.D.
Department of Fisheries and Allied Aquacultures
Auburn University
Auburn, Alabama 36849 USA[117,100,101,46,110,114,117,98,117,97,64,49,101,99,100,121,111,98]
Related Posts

Health & Welfare
10 paths to low productivity and profitability with tilapia in sub-Saharan Africa
Tilapia culture in sub-Saharan Africa suffers from low productivity and profitability. A comprehensive management approach is needed to address the root causes.

Aquafeeds
A look at the SME controlled extrusion process
A study was conducted using a Twin-Screw Extruder equipped with Specific Mechanical Energy (SME) and Density Control valves, to determine the effect of SME on the water stability of shrimp feeds. Further research is needed to evaluate the performance.

Responsibility
A look at unit processes in RAS systems
The ability to maintain adequate oxygen levels can be a limiting factor in carrying capacities for RAS. The amount of oxygen required is largely dictated by the feed rate and length of time waste solids remain within the systems.

Intelligence
A motive, and a market, for farmed fish in Mexico
Boasting ample areas for aquaculture and a robust domestic demand for seafood – not to mention its close proximity to the U.S. market – a land of opportunity lies in Mexico. Fish farming is primed to meet its potential south of the border.

