Compounds active in QS disruption may be alternatives to antibiotics due to efficacy at low concentrations
Early mortality syndrome (EMS) or acute hepatopancreatic necrosis disease (AHPND) is a shrimp disease that has been disrupting production in major shrimp-producing countries since 2009. First reported in China, it has spread to Vietnam, Malaysia and Thailand, and just recently to Mexico and possibly India.
EMS outbreaks typically occur within the first 30 days after stocking newly prepared shrimp ponds, and mortality can exceed 70 percent. EMS is caused by specific strains of a relatively common bacterium, Vibrio parahaemolyticus, which are transmitted orally, colonize the shrimp gastrointestinal tract and produce a toxin that causes tissue destruction and dysfunction of the shrimp digestive organ known as the hepatopancreas.
As reported during sessions on health management at GOAL 2013, the pathogenicity of EMS/AHPND is most likely regulated by a mechanism called quorum sensing, which allows the Vibrio colonies to coordinate the release of the potent toxin.
EMS management
Because EMS is caused by a Vibrio that is difficult to eradicate from aquaculture production environments, its control requires a very different approach than the strategies used against white spot syndrome virus, which are based on specific biosecurity measures. Avoiding early contamination through the broodstock and larval stages, combined with continued control of microbial development – particularly during the initial month of the cycle – will be crucial to control EMS.
In this regard, intensive nursery systems are now being explored to produce juvenile shrimp throughout the critical stages affected by EMS. These systems allow superior control over nutrition and the microbial environment compared to direct stocking into grow-out ponds.
Microbial control
The use of antibiotics to control microbial development throughout the production process is not desirable due to the risk of building up resistance and rejection by legislators and consumers. The shrimp industry requires alternative ways to control the microbial ecosystems in production systems.
Sustainable approaches to modulate the gut microflora in shrimp include the use of probiotics, selected bacteria that inoculate the gut; and specific natural compounds called botanicals or phytobiotics, which are capable of modulating the microflora toward a favorable composition.
Provided botanical formulations are heat stable, they can be easily incorporated into feed at the mill and therefore be present in every meal from the starter feed onwards without requiring major adaptations of the production protocols at nurseries or farms. Phytobiotics that promote healthy gut microflora also support the establishment of probiotic bacteria and therefore enhance the effects of probiotic inoculations in production systems. Various studies have demonstrated the ability of natural products to improve shrimp growth and survival.
Functional feeds
Functional feeds containing gut health promoters allow delivering with every meal an adequate concentration of natural antimicrobial activities into the shrimp gut. These feeds could be an important component of any strategy to prevent EMS. However, the success of this approach will depend on the efficacy of the selected gut health promoter against the pathogenic bacteria involved in EMS.
Synergistic blends of natural compounds can be selected on their bacteriostatic and bactericidal properties against a specific range of pathogenic bacteria in vitro. In work at the Nutriad Technology Center, different Vibrio species, including V. parahaemolyticus, appeared to be highly sensitive to a natural feed additive composed of a synergistic blend of antimicrobial compounds (Table 1).
Coutteau, Efficacy of a natural botanical product, Table 1
| Pathogenic species | Strain | Minimum Inhibitory Concentration (% extract) | Host Range | Quorum Sensing Documented |
|---|---|---|---|---|
| Flavobacterium columnare | LMG 10397 | 0.06 | Tilapia, freshwater fish species | No |
| Listonella anguillarum | LMG 4411 | 0.23 | Most marine fish species | Yes |
| Photobacterium damselae | LMG 7892 | 0.47 | Sea bream, seabass, sole | No |
| Vibrio harveyi | BB120 | 0.47 | Sea bream, common snook, penaeid shrimp | Yes |
| Vibrio alginolyticus | LMG 4409 | 0.94 | Sea bream, grouper, most marine fish | Yes |
| Vibrio parahaemolyticus | LMG 4423 | 0.94 | Marine fish, penaeid shrimp (EMS) | Yes |
| Edwardsiella ictaluri | LMG 7860 | 1.88 | Catfish | Yes |
| Edwardsiella tarda | LMG 2793 | 1.88 | Turbot, tilapia | Yes |
| Pseudomonas fluorescens | DVK1 | 3.75 | Striped bass, white perch, yellow tail | Yes |
| Pseudomonas putida | DVK2 | 3.75 | Ayu, freshwater fish species | Yes |
| Yersinia ruckeri | LMG 3279 | 3.75 | Salmonids, mainly rainbow trout | Yes |
| Aeromonas hydrophila | LMG 2844 | 7.50 | Salmonids, cyprinids, catfish, freshwater fish species | Yes |
| Aeromonas salmonicida | LMG 3780 | 7.50 | Salmonids, cyprinids, freshwater fish | Yes |
| Streptococcus iniae | CCUG 27303 | 8.00% | Trout, tilapia and other freshwater fish | No |
Quorum sensing
Recent research has shown that, apart from direct bactericide/bacteriostatic effects, selected combinations of antimicrobial compounds are at the basis of more complex mechanisms to steer microbiota composition. In human medicine, compounds active in quorum sensing (QS) disruption are being increasingly investigated as potential alternatives to antibiotics due to their efficacy at low concentrations and the low chance of bacteria developing resistance against these non-lethal molecules.
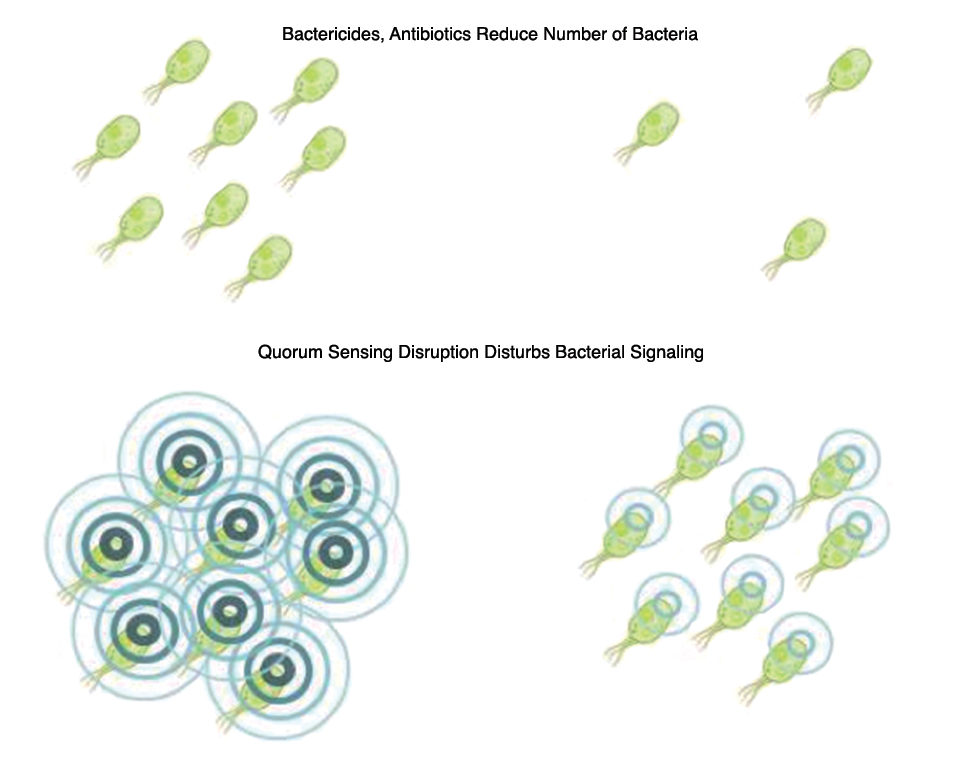
Quorum sensing is a form of bacterial communication based on the production and secretion of signaling molecules that can be detected by adjacent bacteria. When population density rises, these molecules accumulate in the extracellular environment, thereby providing a means for bacteria to quantitatively monitor the presence of other bacteria. When these signaling molecules reach a certain threshold concentration, they initiate intrabacterial signaling that culminates in the activation of specific genes.
Quorum quenching
As determined in work by researcher Dr. Tom Defoirdt and co-workers in 2011, in most pathogenic bacteria for which quorum sensing has been studied, QS was associated with pathogenicity, such as biofilm formation and the production of proteases, invasion factors or other virulence factors. In recent years, research focusing on ways to disturb QS signaling – also called quorum quenching – has therefore gained interest.
Blocking communication is a novel way to prevent bacteria from triggering pathogenicity without exposing them to a selective pressure to survive (Fig. 1). Research by the authors has shown that synergistic blends of natural antimicrobial compounds can function as powerful interrupters of bacterial QS signaling in a typical aquaculture pathogen such as Vibrio harveyi at concentrations well below minimal inhibitory concentrations.
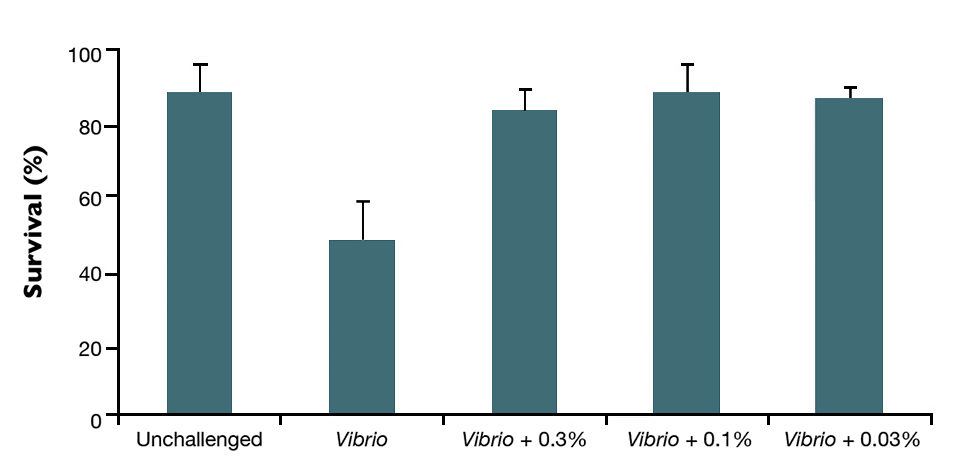
Recent scientific studies by Pande Gde Sasmita Julyantoro and Defoirdt have shown that quorum sensing-disrupting compounds are capable of increasing survival of crustaceans challenged with Vibrio harveyi, including larvae of giant freshwater prawns, Macrobrachium species, and the brine shrimp artemia. Research by this article’s authors similarly showed that strongly diluted extracts from a synergistic botanical product could protect artemia during a challenge with Vibrio harveyi (Fig. 2).
The determination of Vibrio concentrations in varied challenge treatments showed that the strong bactericide effect of the botanical product was responsible for this protection at the highest concentrations of the product. However, the negligible effect on Vibrio concentrations in artemia as well as the culture water in the treatment exposed to the lowest dosage indicated the QS disruption mechanism was responsible for the protective effect of the botanical extract at lower concentrations.
(Editor’s Note: This article was originally published in the January/February 2014 print edition of the Global Aquaculture Advocate.)
Authors
-
Peter Coutteau, Ph.D.
Nutriad International N.V.
Schietstandlaan 2
2300 Turnhout, Belgium[109,111,99,46,100,97,105,114,116,117,110,64,117,97,101,116,116,117,111,99,46,112]
-
Tim Goossens, Ph.D.
Nutriad International N.V.
Schietstandlaan 2
2300 Turnhout, Belgium
Related Posts

Health & Welfare
Beneficial microbes and pathogen control
A range of alternative products is available to improve animal health and water quality, and control pathogen loads. Beneficial microbes produce antimicrobial compounds and suppress pathogen proliferation.

Health & Welfare
Asepsis key to prevent contamination in shrimp hatcheries
Maintaining biosecurity and asepsis in larval shrimp production is a key component of the production chain in Ecuador, which requires the production of 5.5 billion larvae monthly from 300-plus hatcheries.

Health & Welfare
A holistic management approach to EMS
Early Mortality Syndrome has devastated farmed shrimp in Asia and Latin America. With better understanding of the pathogen and the development and improvement of novel strategies, shrimp farmers are now able to better manage the disease.

Health & Welfare
Four AHPND strains identified on Latin American shrimp farms
Two virulence genes are known to encode a binary photorhabdus insect-related toxin that causes acute hepatopancreatic necrosis disease in shrimp. The pathogenicities of these V. campbellii strains were evaluated through laboratory infection and subsequent histological examination in P. vannamei shrimp.


