It’s time to improve testing methodology
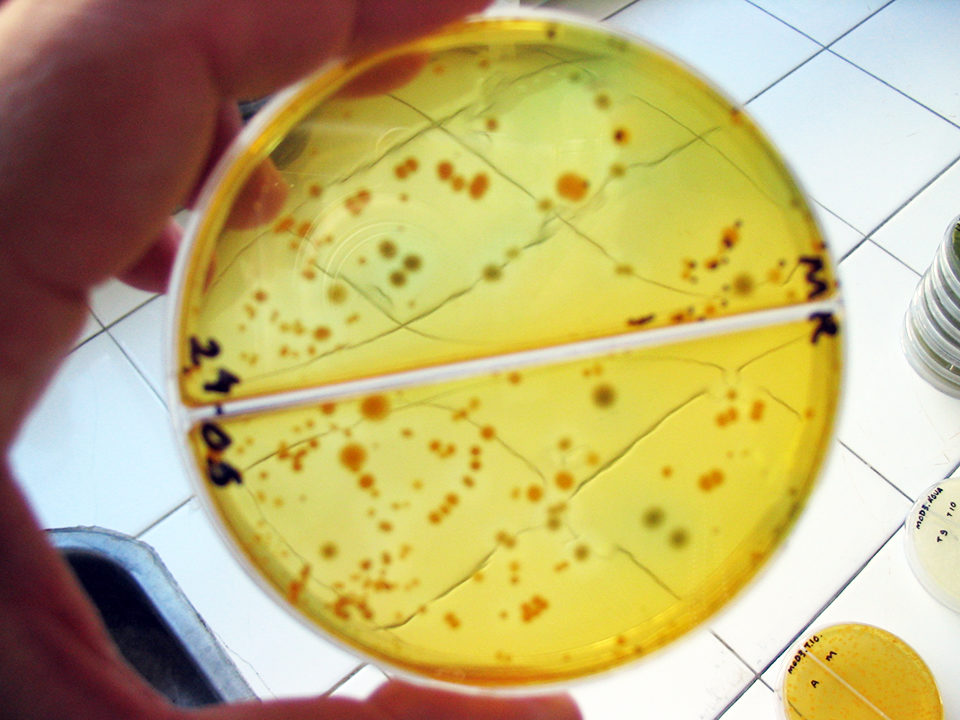
It is tempting to think that antimicrobial resistance (AMR) has evolved only recently in response to the use of antibiotics. That is the general perception. Antibiotics, however, do not play a role in the emergence of resistance, but in its spread.
Emergence of resistance occurs mostly by chance and is independent of the presence of the antibiotic. The antibiotic only provides the positive selective pressure on resistant mutants.
Phylogenetic analysis has shown that this mechanism evolved millions of years ago, indicating the importance of antibiotic resistance genes in bacterial evolution. Lacking this understanding has led to incorrect interpretations in aquaculture’s reported AMR science.
Presence is not evidence
One of the most common misinterpretations is assuming that the detection of AMR is evidence of the use of antimicrobials. As we now understand, when reporting the presence of resistance isolates, detection of the increase in resistance to a particular compound should be reported rather than simply the presence of isolates.
Controls needed
A large number of studies have reported significant to high levels of resistance in the microflora of relatively pristine, unpolluted water or water bodies free of known anthropogenic influence. Others have identified the increase of resistance frequency in environments impacted by an aquaculture facility but not by any antimicrobial agent used in the facility.
Furthermore, a dramatic increase of resistance in environments where there is significant nutrient enrichment has also been documented. For example, in laboratory mesocosm, environmental samples can increase their resistance frequency to oxytetracycline from 2 to 100 percent purely by their incubation in the presence of drug-free fish feed.
Temporal and spatial controls of the background resistance frequencies are absolutely necessary. If adequate controls are lacking, the data presented in studies cannot be treated as contributing to our understanding of this issue.
Methodology problems
There are no standard methods for the determination of resistance frequencies. The use of different methods, media and breakpoint concentrations in different studies has the consequence that little or no comparison can be made of the available data.
The initial step toward universal standard test protocols was taken by a group of 24 international scientists who met in the 1990s. The current disk diffusion protocols are presented in Clinical and Laboratory Standards Institute document M42-A, and the minimum inhibitory concentration protocols are in M49-A. Adherence to standard protocols does not guarantee that all laboratories will generate quantitative data that is absolutely comparable. Interlaboratory variation still remains a problem.
Recommendations
Aquaculture’s scientific community needs to improve its understanding of AMR processes to provide the best interpretations of its work and result in correct decisions. Improving the methodology of AMR testing, the obligatory use of temporal and spatial controls, and the implementation of laboratory ring tests are the first steps to start producing reliable information.
(Editor’s Note: This article was originally published in the November/December 2009 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Victoria Alday-Sanz, Ph.D.
Gran Via 658, 4-1 08010
Barcelona, Spain[109,111,99,46,111,111,104,97,121,64,121,97,100,108,97,95,97,105,114,111,116,99,105,118]
Tagged With
Related Posts

Innovation & Investment
American Unagi brings eel farming back ‘home’
Sara Rademaker launched American Unagi to shift eel farming to American soil, where the eels are from. Why? Because of the novelty, and because she saw an opportunity to do things better.

Aquafeeds
Animal co-product hydrolysates sources of key molecules in aquafeeds
Key molecules found in animal byproduct hydrolysates show potential for use as functional ingredients in aquaculture feeds. Animal co-product hydrolysates from slaughterhouse waste and rendered animal byproducts present a protein alternative.

Health & Welfare
Antibiotic-resistant bacteria, part 1
No antimicrobial agent has been developed specifically for aquaculture applications. However, some antibiotic products used to treat humans or land-based animals have been approved for use at aquaculture facilities.

Health & Welfare
Antibiotic-resistant bacteria, part 2
The development of antimicrobial resistance genes in human pathogens as a consequence of exposure to antibiotics in aquaculture is widely documented. Reports implicate foodborne antibacterial-resistant pathogenic bacteria in human disease.


