Performance of lined, intensive ponds compared with extensive earthen ponds

Shrimp culture in Panama virtually came to a halt in 1999 and 2000 due to the White Spot Syndrome Virus and, to a lesser extent, Infectious Hypodermal and Hematopoietic Virus, and Necrotizing Hepatopancreatitis. Like Ecuador, where production in 2000 fell to approximately 20 percent of pre-1999 levels, Panama has experienced widespread bankruptcy and idled ponds.
To address these disease problems, an agreement between the Texas Agricultural Experiment Station (USA) and Agromarina de Panama, the oldest shrimp farm in the Western Hemisphere, led to a test during the second half of 2000. At Agromarina de Panama (owned by North American Agrisystems, Inc. of Houston, Texas, USA), new nursery and grow-out ponds were built with liners for intensive production beside concurrently stocked, existing extensive earthen ponds to compare the performance of the two systems.
Pathogen exclusion a priority

A broad range of potential vectors/hosts has been implicated in WSSV infectivity, so this test was designed to exclude pathogens. All water was filtered to 25 μ (90 percent nominal). Pond liners were used to exclude soil-borne sources. Fences of staked polyethylene were placed around the perimeter for crab control. Additionally, the locally sourced (Litopenaeus vannamei) postlarvae – from wildcollected broodstock that matured and spawned in captivity at the Agromarina de Panama hatchery in Veracruz, Panama – were compared with postlarvae (PL) derived from domesticated stocks of the U.S. Marine Shrimp Farming Program, U.S. Department of Agriculture.
Water circulation and dissolved oxygen were maintained at high levels 24 hours per day to help maintain healthy, nonstressful pond conditions. Temperature, salinity, alkalinity, and pH were maintained at moderate levels. No attempt was made to cover the ponds, so potential aerial sources (avian, for example) were not excluded.
Description of ponds
Adjacent to 920 ha of existing earthen ponds at Agromarina de Panama’s Aguadulce Farm, 18 lined nursery ponds and 18 lined grow-out ponds were constructed (Figs. 1, 2).
Nursery ponds
Rectangular nursery ponds measured 30 x 13 meters (.04 ha) with average depth of 1.5 meters. They were equipped with dual central drains and lined with black, 30-mil (0.8 mm) high-density polyethylene liners. The drain used for animal transfer to grow-out ponds was 30.5-cm-diameter PVC. Aeration/circulation was provided 24 hours per day by two or three, 1.5-hp units from Italy that aspirate surface air to the submerged motor and propeller. Zero water exchange was used, although rainfall provided periodic additions to the ponds
Nurseries were stocked with PL40-60 held for approximately 45 days in raceways at another location to verify no disease expression prior to stocking. PL were stocked in nursery ponds at 274 to 371 per square meter. Local 45 percent-protein crumble feed was distributed manually to ponds four times daily. Growth was determined weekly by checking 30 to 50 individuals sampled by cast-netting from ponds.
A molasses-grain mixture was added to both nursery and grow-out ponds weekly to provide additional carbon substrate for bacteria. Two to three applications of a nitrogen- phosphorus fertilizer were made three days prior to stocking nursery ponds. In both nursery and grow-out ponds, organic sludge was removed weekly through the screened central drain by opening a valve outside the pond.
Grow-out ponds
Grow-out ponds were 0.1 ha square, and also utilized screened, 30.5-cm central drains. Aeration (40 horsepower per hectare, or 4 hp total for each pond) was provided 24 hectares per day from four, 1-hp paddlewheels or two, 2-hp paddlewheels. The design criterion for scaling and positioning of aeration/circulation units in both nursery and grow-out ponds was 6.1 to 12.2 centimeters per second water current within 2 meters of the perimeter and zero velocity around the central drain.
Juveniles were held in the nursery ponds for six to eight weeks prior to transfer by gravity drain into the grow-out ponds. Assuming 90 percent survival through the nursery ponds, stocking density of grow-out ponds at the time of transfer was 123 to 130 per square meter. Feed (35 percent protein with 3.5 percent squid meal) was distributed manually four times daily, as in the nursery ponds. Two feed trays per pond were routinely monitored for consumption and corresponding feed rate adjustment.
When excessive phytoplankton blooms occurred, calcium hydroxide was added to cause rapid decline of the bloom. Alkalinity was maintained at 100-120 milligrams per liter throughout the cycle with periodic additions of calcium carbonate.
Control ponds
Eleven earthen extensive ponds, which served as controls for comparison with the new intensive ponds, werestocked with postlarvae from the Agromarina hatchery. The PLs came from wild broodstock matured in captivity. Extensive ponds ranged in size from 2.2 ha to 20.2 ha. Postlarvae ranged from PL12 to PL19 at stocking. Stocking density ranged 2.7 to 4.3 per square meter. Half of the ponds were unfed, and the other half were fed a 20 to 25 percent protein pelleted diet.
Water source

Seawater used for both the intensive and extensive ponds came from the estuary that bisects the farm, approximately two km from the Gulf of Parita, Pacific Ocean. For the extensive ponds, seawater was filtered to 285 μ from the farm’s water distribution canal.
For the new intensive ponds, seawater from the canal was pumped through a pressurized 25-μ mixed-media filter into a temporary holding pond, then directly to the culture ponds prior to stocking. No seawater was added to the intensive ponds throughout the nursery and grow-out culture, although periodic rainfall did occur. Likewise, in the extensive ponds, no water exchange was used after filling.
Water quality
Dissolved oxygen was monitored every two hours, and pH, twice daily. Daily oxygen values ranged from 5 to 8 ppm, and never dropped below 4.5 ppm. Daily temperature range was 26 to 31 degrees-C. Weekly samples were taken for salinity, alkalinity, phosphorus, ammonia, nitrite, nitrate and zooplankton and phytoplankton enumeration. Harvest was accomplished through central drains by gravity, with some assistance from workers (Fig. 3).
Intensive vs. extensive ponds
Results of the comparison between the pilot lined ponds and the conventional earthen ponds were dramatic (Table 1). In the 12 lined ponds, average survival was 80 percent (range 62 to 93 percent), following 125 days of culture in nursery and grow-out ponds, while average survival was 8.7 percent (range 4 to 20.3 percent) after only 59 to 83 days of culture in the 11 concurrently operated extensive ponds.
Bray, Harvest summary for lined and earthen test ponds, Table 1
| Treatment | Number Of Ponds | Pond Size (ha) | Days | Stocking Density | Average % Survival | Average kg/ha | Average Weight (g) | Average Growth/wk (g) |
|---|---|---|---|---|---|---|---|---|
| Intensive Ponds, Lined Wild Broodstock PL40-60 | 12 | 0.1 | 125 | 123/m2 | 80 | 11,086 | 10.2 | .57 |
| Intensive Ponds, Lined Domestic Stock PL40-60 | 6 | 0.1 | 101 | 130/m2 | 80 | 13,241 | 11.8 | .81 |
| Extensive Ponds, Earthen Wild Broodstock PL12-19 | 11 | 2.2-20.2 | 59-83 | 2.7-4.3/m2 | 8.9 | 24 | 8.7 | 1.12 |
Clinical signs of WSSV were not observed in the intensive ponds at any of the weekly sampling periods, while in the extensive ponds, all ponds experienced two to three episodes of WSSV die-off. An average of 23.6 kilograms per hectare per crop was achieved in the earthen ponds, compared with an average 11,086 kilograms per hectare in the intensive ponds.
It is unclear whether the higher survival in lined ponds was due to the use of nursed postlarvae, pond liners, finer filtration of water, perimeter crab barriers, or a combination of the above factors.
Domesticated vs. wild broodstock
In the lined ponds, no difference in survival at harvest was observed between the domesticated PL source (six ponds) and the wild broodstock-derived PL (12 ponds). Both groups averaged 80 percent survival (range 70 to 94 percent in the domesticated PL ponds, 62 to 93 percent in the wild broodstock-derived PL ponds). However, average growth per week was 42.1 percent higher in the domesticated PL ponds for the entire period from nursery stocking to grow-out pond harvest.
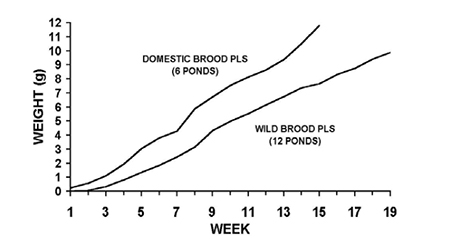
This resulted in higher average harvest biomass per ha (13,241 kilograms per hectare in the domesticated PL ponds versus 11,086 kilograms per hectare in the wild broodstock-derived PL ponds), even though the wild broodstock PL ponds were extended, on average, 24 additional days. Additionally, shrimp harvested from the domesticated stock PL were uniform in size. Runt deformity syndrome, indicative of IHHN virus, was observed in all ponds stocked with wild broodstock-derived PL, although no mortality due to this condition was observed. Fig. 5 compares average growth per week between domesticated PL ponds and wild broodstock-derived PL. The growth difference in the populations was clear from early in the trial (Fig. 4).
Results achieved in the earthern test ponds were consistent with the entire farm’s recent production. During the 30 days prior to harvest of the intensive ponds, 462 ha of earthen ponds were harvested, with average survival of 8.34 percent and average production of 24.3 kilograms per hectare.
Effect of soil
An additional observation from the Agromarina de Panama farm added evidence that pond soil condition affects WSSV incidence. Nine small earthen ponds (0.1 to 0.4 ha, mostly 0.2 ha) were stocked at around 8 per square meter. In four ponds, bottoms were renovated prior to stocking. The upper layer of soil was actually removed from the ponds.
In these ponds, average survival was 78.5 percent (range 52 to 100 percent). In the other five small ponds, bottoms were untouched for the previous two to three growing cycles, except for drying between crops. In these ponds, average survival was only 42.4 percent (range 24 to 55 percent).
There was one additional variable in the bottom-scraped ponds. Calcium carbonate was added at a rate of 45 kilograms per hectare biweekly. Nonetheless, alkalinity and pH measurements were not different between the two groups, suggesting the calcium carbonate addition may not have been related to the survival difference observed. This comparison seems to indicate that a soil-borne source of WSSV could be a primary issue in management of this virus.
Conclusion
These results indicate it is possible to reestablish L. vannamei production in a WSSV-plagued region, even with a continued viral source on the same farm. Simple exclusion of potential vectors via postlarvae, water, and soil, along with management techniques to maximize water quality, allowed normal culture of L. vannamei to proceed again in a contaminated area.
Additionally, the increased growth potential of diseasefree, genetically selected stocks – even in a virus-contaminated environment – was demonstrated.
(Editor’s Note: This article was originally published in the February 2001 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
William A. Bray, M.S.
Senior Research Associate
Texas Agricultural Experiment Station
Texas A&M University System
Port Aransas, Texas USA[109,111,99,46,108,111,97,64,97,119,121,97,114,98]
-
Addison L. Lawrence, Ph.D.
Regents Professor, Faculty Fellow
Texas Agricultural Experiment Station
Texas A&M University System
Port Aransas, Texas USA[109,111,99,46,111,111,104,97,121,64,108,108,97,112,109,115]
-
Melissa del C. Royo
Manager, Pilot Intensive Pond Project
Agromarina de Panama, S.A.
Aguadulce, Panama[109,111,99,46,108,105,97,109,116,111,104,64,97,100,111,112,111,116,99,111]
-
William R. More
Vice President, Operations
Agromarina de Panama, S.A.
Aguadulce, Panama[116,101,110,46,111,99,114,101,99,64,49,101,114,111,109,114,119]
Tagged With
Related Posts

Health & Welfare
Botanical extracts improve productivity of shrimp, pangasius
In a study with pangasius, dietary administration of a blend of botanical extracts improved performance, reducing intensity of gill parasite infestation.

Health & Welfare
CENIACUA develops WSSV-resistant shrimp in Colombia
To combat white spot syndrome virus (WSSV) in white shrimp, Corporación Centro de Investigación de la Acuacultura de Colombia (CENIACUA) initiated a selective-breeding program to develop resistance in shrimp.
Health & Welfare
Double-stranded RNA against WSSV genes provides antiviral protection in shrimp
Silencing genes in white spot syndrome virus (WSSV) with critical roles in replication could provide a strong antiviral effect and thus reduce shrimp mortality. The authors therefore established a study to evaluate the antiviral efficacy of double-stranded (ds)RNA against non-structural WSSV genes.

Health & Welfare
Ecuador study shows wild postlarvae use down
During the 1970s and 1980s, Ecuador's shrimp industry relied almost entirely on wild postlarvae but has since developed large hatcheries.


