Examining the use of prebiotics, probiotics and lipopolysaccharides

Antibiotics have traditionally been used to treat microbial infections in aquaculture around the world. However, the use and abuse of antibiotics can cause unwanted adverse effects such as the accumulation of antibiotics in the tissues of animals, immune suppression, development of bacteria resistant to antibiotics and destruction of environmental microbial flora.
Therefore, research aimed at replacing antibiotics with natural products such as probiotics, prebiotics and lipopolysaccharides (LPS) has received considerable attention, ensuring the development of different bioprocesses to obtain these molecules.
The term probiotic means “for life,” and is derived from the Greek words “pro” and “bios.” The widest definition of probiotics was formulated by Fuller, who indicated that a probiotic is considered as “a live microbial food supplement that beneficially affects the host animal by improving its intestinal balance.” However, not only live microorganisms should be considered as probiotics, but also any microorganism or any of its components that can confer any beneficial physiological effect to the host or the environment where the microorganism lives.
A prebiotic, unlike a probiotic, is not an organism and therefore has less influence on the natural environment. Based on the definition of Gibson and Roberfroid, prebiotics are non-digestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of one or a limited number of bacteria in the colon, thus improving the health of the host.
Prebiotics have two main characteristics: They are natural foods, and they are not living organisms, so this fact constitutes an advantage over probiotics. The incorporation of prebiotics in the diet does not need authorization as food additives because they are well described in the literature. Originally, prebiotics were used to stimulate bifidobacteria and lactobacilli in the human microbiota. Lipopolysaccharides induce strong effects on the immune system of shrimp.
This review article – adapted from Aquacultura (Ecuador) No. 121, Feb. 2018 – highlights the use of probiotics, prebiotics and LPS, mechanisms of action and future challenges in the use and production of these molecules in aquaculture, including shrimp culture.
Probiotics in aquaculture
Probiotics are cultured microbial formulations that are mixed with enzymes and fermented cereals attached to a substrate or formulation for use in aquaculture. In the field of aquatic organisms, probiotics have been defined as “microbial cells capable of traveling through the gastrointestinal tract and staying alive, with the aim of improving health.”
A broader definition postulates that “probiotics are defined as living or dead entities, or even a component of microorganisms that act under different modes of action by conferring beneficial effects not only to the host but also to their environment.” Table 1 shows microbial strains successfully tested as probiotics under field conditions. There is a growing interest in probiotics that has been stimulated by several factors, including:
1) Interesting scientific and clinical results using well documented probiotic organisms.
2) Concerns about side effects and environmental damage.
3) The use of natural products.
4) The development of interesting aquatic antagonists for organic aquaculture.
Trujillo, farmed shrimp, Table 1
| Probiotic strain | Benefit | Application mode | Reference |
|---|---|---|---|
| Bacillus cereus | Control over vibrio concentration | In water | Moreira D, Sabrina M, Leivas FP, Romano LA, Ballester E. New Bacillus probiotic tested for shrimp. GAA Advocate 2011 -Jan/Feb: 46-47. |
| Bacillus licheniformis | Competes with other bacteria for organic matter accumulation and cleaning | In water | Moriarty DJW, Decamp OP, Lavens P. Probiotics in Aquaculture. AQUA Cult. Asia Pacific Mag. 205; 1:14-16. |
| Bacillus subtilis | Against infection by Vibrio harveyi | Added to water | Utiswannakul P. Sangchai S, Rengpipat S. Enhanced growth of black tiger shrimp, Penaeus monodon, by dietary supplementation with Bacillus (BP11) as a probiotic. J. Aqua. Res. Dev. 2011 - 3:2155-2164. |
| Saccharomyces cerevisiae | Immunostimulation and protection against Vibrio harveyi | In the diet | Scholtz U, Garcia Diaz G, Ricque D, Cruz Suarez LE, Vargas Albores F, Latchford J. Enhancement of vibriosis resistance in juvenile Penaeus vannamei by supplementation of diets with different yeast products. Aquaculture 1999; 176 (3-4): 271-283. |
| Streptococcus sp. | Antagonist to Vibrio alginolyticus | In the diet | Ajitha S, Sridhar M, Sridhar N, Singh ISB, Varghese V. Probiotic effects of lactic acid bacteria against Vibrio alginolyticus in Penaeus (Fenneropenaeus) indicus. Asian J. Fish. Sci. 2004; 17:71-80. |
| Pediococcus acidilactici | Control of Vibrio alginolyticus infection | In the diet | Villamil L, Figueras A, Planas M, Novoa B. Control of Vibrio alginolyticus in Artemia culture by treatment with bacterial probiotics. Aquaculture 2003; 219(1-4):43-56. |
Probiotics work according to competitive exclusion mechanisms by which pathogens are replaced or excluded by the development of a beneficial microbial population on the intestinal surface, which reduces the incidences of the disease, and promotes better health and better growth of the host (Fig. 1). Bacterial strains or their components, are capable of producing inhibitory substances that could interact directly with bacterial and viral pathogens.
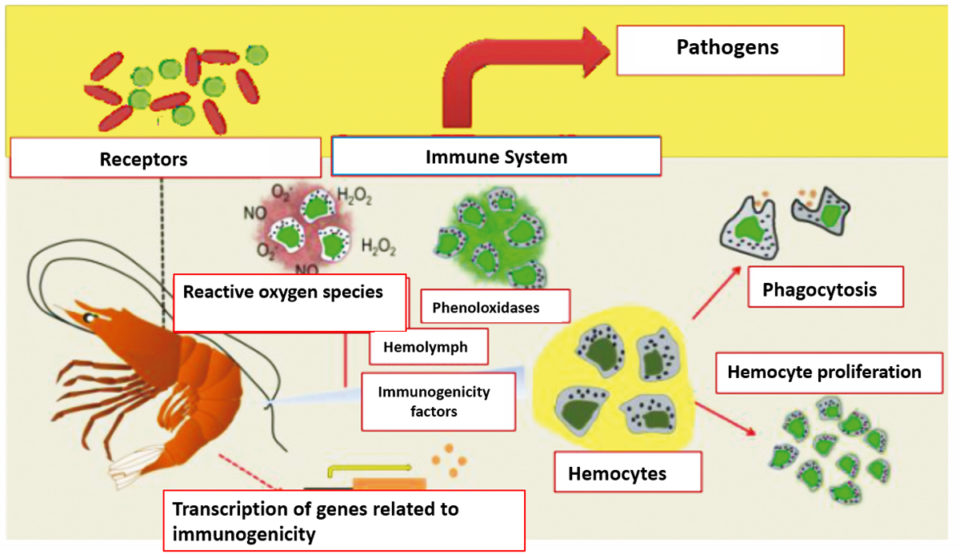
The growth of these pathogens is prevented through the production of antagonistic metabolites or by adhesion interference. Some of the previous studies conducted in the first 90 years of the last century, reported by Nogami and Maeda, and by Rengpipat et al., showed better growth and survival of crustaceans with probiotic supplements. Some studies describe a significant increase in the weight of shrimp treated with probiotics over the untreated group at the farm level as shown in Table 2.
Trujillo, farmed shrimp, Table 2
| Parameter | Tanks treated with probiotics (g) | Tanks not treated with probiotics (g) |
|---|---|---|
| Initial weight | 126.6±1.1 | 127±1 |
| Final weight | 160.7±3.4 | 309±2.3 |
| Weight gain | 234.1±2.6 | 182±1.3 |
| Mean initial weight | 4.22±0.11 | 4.23±0.03 |
| Mean final weight | 12.02±0.35 | 10.3±0.05 |
| Mean weight gain | 7.8±0.09 | 6.07±0.02 |
| Survival (%) | 100 | 100 |
Lara-Flores et al. explained that probiotic bacteria are good candidates for increasing nutrient digestion and growth in aquatic organisms. Irianto and Austin stated that the harmful compounds in the diet can be detoxified by the action of hydrolytic enzymes such as amylases and proteases, and also that nutrients in organisms could be improved by vitamins such as biotin and vitamin B12.
Digestive enzymes are necessary to break down complex compounds into simpler, more absorbable molecules that could be used by the host. Therefore, dietary supplementation with probiotics increases growth through the enzymatic contribution that increases digestibility.
The constituents of the digestion of proteins generated by the action of endogenous proteolytic enzymes also contribute to increase the digestibility of the feed associated with the administration of probiotics. In fact, the probiotic bacterium Lactobacillus plantarum improves gene expression and immune responses in the Pacific white shrimp (Litopenaeus vannamei) when combined in its diet. Specifically, L. plantarum improves the activity of phenoloxidase, prophenoloxidase (proPO) and superoxide dismutase (SOD), together with the survival rate after stimulation with Vibrio alginolyticus.
These effects are observed when bacteria are given in the diet at 1010 CFU/kg for 168 hours. L. plantarum is highly effective against Vibrio harveyi. Experimental challenges in vivo, shows that L. vannamei increased resistance compared to a control group not exposed to V. harveyi. These results have suggested that the probiotic strain has an immunoreactive effect on the host, possibly related to extracellular compounds released from bacteria that can cause nonspecific immune responses.
The activity of L. plantarum is responsible for the increase in the total hemocyte count and the PO activity. According to the results of this research, it was suggested that the binding activity is the main cause related to the most effective elimination of hemolymph and hepatopancreas by shrimp fed with the diet supplemented with probiotic.
When challenged with Vibrio nigripulchritudo, Pediococcus acidilactici influenced the antioxidant defenses and oxidative stress of L. vannamei. It shows efficacy in antioxidant defense markers such as SOD, catalase, glutathione peroxidase, glutathione and tissue damage. This strain was efficient in maintaining antioxidant defense levels for a longer period than the control and non-infected shrimp groups.
Among the three strains, Bacillus P64 shows a significantly higher immunity index and an immune response similar to that of V. alginolyticus. In addition, it induces an increase in the population of hyaline cells. These strains show antagonistic activity against shrimp pathogens. When juvenile shrimp are fed chitin and Vibrio gazogenes, the number of vibrio type bacteria in the anterior and posterior gut decreases significantly and the number of hemocytes changes significantly.
The change in the number of hemocytes probably reflects the immune status of shrimp because hemocytes are involved in both cellular and humoral defenses of shrimp. Yeast β-glucans have also been applied as probiotics in aquaculture to modulate the innate immune system of fish and to improve the survival of fish against pathogens.
Prebiotics in aquaculture
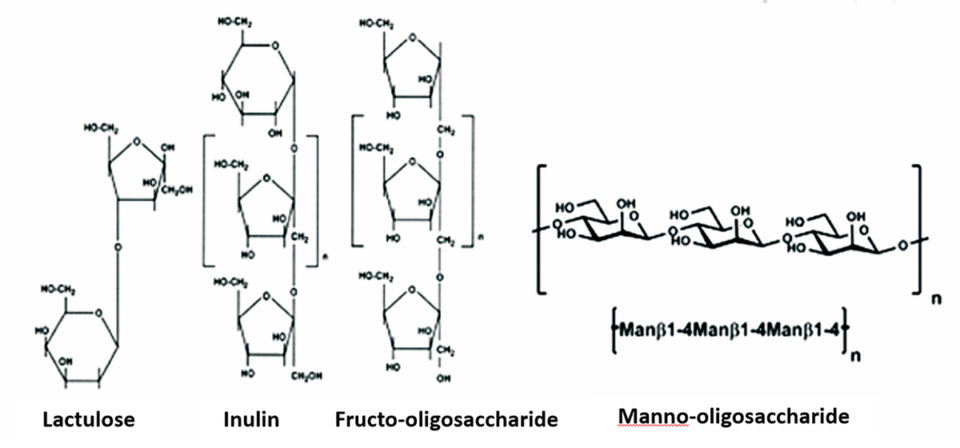
Prebiotics, unlike probiotics, are not living organisms so they have less influence on the environment. Mahious et al., Fooks et al. and Gibson et al. described that any food that reaches the colon – for example, non-digestible carbohydrates, peptides, proteins and certain lipids – can be a candidate for prebiotic. However, non-digestible carbohydrates such as oligosaccharides, inulin, transgalactooligosaccharide and lactulose are considered the focus of most oligosaccharides.
In fish, the mannan oligosaccharides (MOS) and fructo-oligosaccharides (FOS) are perhaps the most studied oligosaccharides despite information on the use of other oligosaccharide-like compounds in aquatic animals. The chemical structure of some of these oligosaccharides is presented in Fig. 2. Some studies have shown that prebiotics can modify the microbial community of the gastrointestinal tract to enhance non-specific immune responses. The prebiotic fermentation in the colon by the bacteria causes a significant modification of the colonic microflora.
In fact, oligosaccharides serve as a substrate for the growth and proliferation of anaerobic bacteria, mainly bifidobacteria, which inhibit the growth of putrefactive and pathogenic bacteria present in the colon. The resulting production of substances that stimulate the immune system increases the protection of the host against infections.
Li et al. found that supplementation with short-chain FOS at concentrations of 0.025 to 0.8 percent (m/m) increased the respiratory capacity of hemocytes in Pacific white shrimp (L. vannamei) grown in a recirculated system.
Prebiotics can modify the microbial community of the gastrointestinal tract to improve non-specific immune responses. They also help reduce the pH of the intestine through the formation of short-chain fatty acids, which favors the beneficial bacteria that block the adhesion of the pathogens and sites of toxin receptors, nutrient competition and stimulation of the immune system. However, reports on the effect of prebiotics on the growth parameters of the species are inconclusive and some are contradictory.
The Beluga sturgeon diet (Huso huso) supplemented with 1, 2 and 3 percent (m/m) of inulin has adversely affected a variety of parameters such as weight gain, growth, protein efficiency ratio, protein and energy retention, and food efficiency. In addition, Refstie et al. found that Atlantic salmon fed for three weeks with a diet based on fishmeal supplemented with 75 g/kg of inulin increased the relative mass of the gastrointestinal tract, but the absorption capacity of the fish is not affected. A diet with 20 g/kg of oligofructose, a FOS produced by partial enzymatic hydrolysis of inulin, increases the growth of turbot larvae, but 20 g/kg of inulin itself has no effect on growth.
The intake of prebiotics depends on the type of ingredients used in the formulations of the diet and, therefore, will vary between the species and the feed supplied to the animals. The type of prebiotic to be supplemented, specific animal characteristics (species, age, stage of production) and type of diet have been identified as important considerations when supplementing prebiotics in fish diets.
However, many experimental results have not been conclusive regarding the benefit of some of these prebiotics. And attention must be paid to the development of bioprocesses, practical formulations and economic considerations that ensure sustainable production.
Lipopolysaccharides as immunostimulators
The effects of lipopolysaccharides (LPS) on the immune system of shrimp have been the subject of numerous articles. Interesting cases that can provide evidence of the possible application of LPS as an immunostimulant in shrimp farming are described below. The percentages of proliferation of hemocytes can increase approximately three times after stimulation with LPS. In addition, uptake of 3 H thymidine in circulating hemocytes can be significantly 26 times higher in shrimp stimulated with LPS than in unstimulated ones.
The proPO activation system in Penaeus spp. improves greatly with the LPS treatment, which suggests its participation in non-self-recognition. In addition, the PO activity in the shrimp serum increased with the LPS treatment.
The oral administration of Pantoea agglomerans with LPS improves disease resistance against penaeid acute viremia and induces the inactivating activity of the viruses in the hemolymph of Kuruma shrimp, Penaeus japonicus. The proliferation of most cells in the hematopoietic tissue of black tiger shrimp (P. monodon) can be significantly increased after LPS injection.
Injection of Pacific white shrimp, L. vannamei, with LPS decreased mRNA levels of antimicrobial peptides, penaeidin 2, penaeidin 3, penaeidin 4 in a dose-dependent manner, while mRNA levels of serine proteinase and proPO did not change significantly.
The stimulation of white shrimp (L. vannamei) hemocytes with LPS leads to the formation of characteristic extracellular traps of fibers made from the DNA of the host cell. The groups of black tiger shrimp (Penaeus monodon) fed with LPS-coated pellets showed higher survival rates than the control groups when challenged against Vibrio harveyi.
In addition, the levels of genes related to immunity such as, for example, the isoform 3 of the anti-LPS factor, the C-type lectin and the mucin-type peritrophin were regulated in the intestines of the P. monodon group fed an LPS-supplemented diet once or twice a day, unlike the control group. The expression of type C lectin 3 in gills of L. vannamei gills is positively regulated after the challenge against LPS, and the LPSs can regulate L. vannamei cactin in hemocytes.
Natural strategies to improve growth and health in large-scale shrimp culture prevent necrosis and release endogenous molecules such as L, β and P. These endogenous molecules can cause changes in cell viability, degranulation and hemocyte necrosis, indicative of activation of in vitro immunity.
Injection of LPS causes a decrease in the total counts of semi-granular and granular hemocytes of P. monodon. In addition, nonspecific esterase activity, the production of reactive oxygen species and the production of nitric oxide are significantly induced in shrimp hemocytes injected with LPS, while the ratio of apoptotic cells to hemocytes increases. Methods for isolating and characterizing LPS have recently been reported for use in shrimp culture.
Perspectives
The application of probiotics, prebiotics and LPS in aquaculture has shown positive results, but the evaluation of the biological effects of these entities in the natural environment and the profitability of the treatment options is insufficient. In addition, the mixing of these biological reagents to form symbiotics has a very promising field in shrimp culture since this strategy has had some success in other marine species.
Following the numerous genome sequencing tools currently in use, future research on probiotics, prebiotics and LPS effects should involve transcriptome and proteome analysis using high throughput assays.
In addition, the development of transcriptomes and proteomes in the gut microbiota should be studied. From the bacteriological point of view, it is of great importance to continue evaluating if the microbial changes have any positive effect on animal health, as they contribute to inhibit the intestine colonization by pathogenic bacteria and improve the innate immunity. Therefore, studies with all these substances should have a high priority in the future, and challenge studies should be included as a gold standard to assess their effects on shrimp health.
References available from first author.
Authors
-
Luis E. Trujillo
Universidad de las Fuerzas Armadas ESPE
Life Science Department
Industrial Biotechnology and Bioproducts Research Group
Center for Nanotechnology and Nanosciences CENCINAT
Quito, Pichincha, Ecuador[99,101,46,117,100,101,46,101,112,115,101,64,51,111,108,108,105,106,117,114,116,101,108]
-
Leonor Rivera
Universidad Técnica de Machala
Machala, El Oro, Ecuador;
and Unidad de Posgrado de la Universidad Nacional Mayor de San Marcos-Peru -
Eugenio Hardy
Universidad de las Fuerzas Armadas ESPE
Life Science Department
Industrial Biotechnology and Bioproducts Research Group
Center for Nanotechnology and Nanosciences CENCINAT
Quito, Pichincha, Ecuador
and Biotechnology Laboratory, Study Center for Research and Biological Evaluations
Institute of Pharmacy and Foods
Havana University, Cuba -
Erika M. Llumiquinga
Universidad de las Fuerzas Armadas ESPE
Life Science Department
Industrial Biotechnology and Bioproducts Research Group
Center for Nanotechnology and Nanosciences CENCINAT
Quito, Pichincha, Ecuador -
Francisco Garrido
Universidad de las Fuerzas Armadas ESPE
Life Science Department
Industrial Biotechnology and Bioproducts Research Group
Center for Nanotechnology and Nanosciences CENCINAT
Quito, Pichincha, Ecuador -
Jesús A. Chávez
Unidad de Posgrado de la Universidad Nacional Mayor de San Marcos, Perú
-
Victor H. Abril
Universidad de las Fuerzas Armadas ESPE
Life Science Department
Industrial Biotechnology and Bioproducts Research Group
Center for Nanotechnology and Nanosciences CENCINAT
Quito, Pichincha, Ecuador -
José M. País-Chanfrau
Universidad Tecnica del Norte UTN. FICAYA.
Ave. 17 julio 5-21, Ibarra 119
Imbabura, Ecuador
Tagged With
Related Posts

Health & Welfare
Prebiotics and their role in sustainable aquaculture
Results of a study to evaluate a commercial prebiotic on growth of European sea bass juveniles indicates this product can improve fish growth performance and allowing for the fish to reach the marketable size earlier, boosting their immune status, and reducing potential fish losses due to pathogens.

Health & Welfare
Focus on gut health increases aquaculture productivity
Increased attention to nutrient digestibility and gut health are two important focus areas for technological development of manufactured feeds in the aquaculture industry. Both laboratory and field studies have demonstrated the potential benefits for aquaculture productivity and the economics of specific feed additives developed to stimulate the establishment of a healthy gut microflora.

Aquafeeds
Biosecurity protocols needed for shrimp feeds, feeding practices
Shrimp aquafeeds – live, fresh or formulated – should not be an entry point of potential pathogens to the shrimp and/or to their culture systems.

Health & Welfare
Biofloc technology holds potential for carnivorous fish species
Juvenile carnivorous African catfish performed well in biofloc-based systems, which could help produce better quality and more disease-resistant seed of this important aquaculture species and support the expansion of African catfish farming industry.


