Stocking into ponds with pH greater than 8.0 can result in mortalities

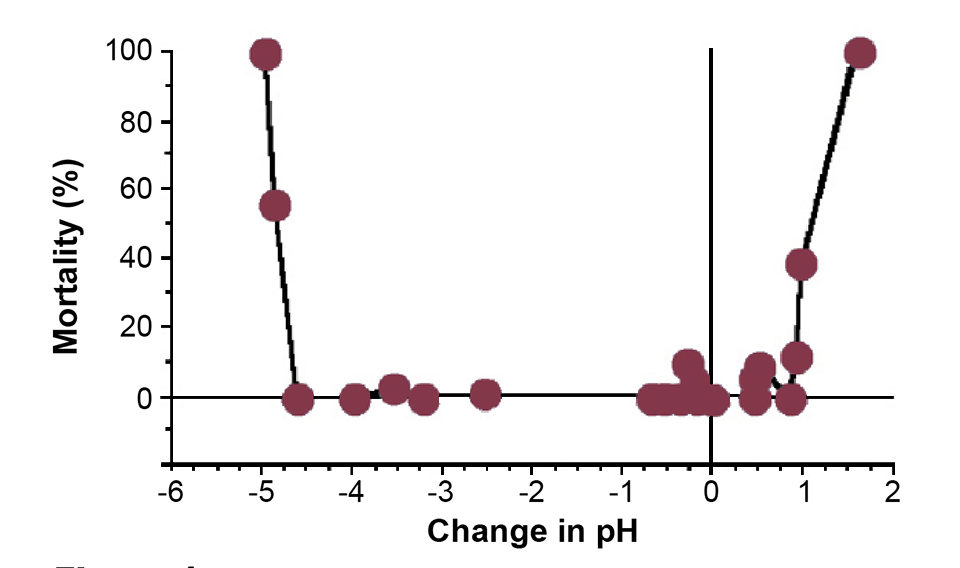
Recent research has shown that seven to fourteen days posthatch, channel catfish fry suffer large mortalities if subjected to an abrupt rise in pH of 0.5 (Fig. 1). In order to assess the implications of this condition for hatchery management, the authors carried out a study were pH values were measured in commercial hatcheries and a variety of ponds into which fry were stocked during the 2007 hatchery season in Mississippi, USA.
Water conditions
Most commercial catfish hatcheries in Mississippi use groundwater supplied from a deep aquifer. These wells range 200 to 400 meters deep in most cases. They are typically moderately alkaline 150 to 200 mg/l and extremely soft with no iron. Calcium chloride is added to the incoming water by metered pumps to raise hardness from near 0 to 50 mg/l.
In the hatchery tanks and troughs containing 4- to 6-day posthatch fry, pH ranged 7.2 to 8.6. Most commonly, pH values were 7.4 to 7.9 in troughs containing 400 to 600 fry/l with water flow rates near 12 l/minute. Values for pH in ponds prepared for receiving fry were commonly 7.8 to 8.2.
Fry transport
However, when fry are loaded into transport tanks, they commonly experience densities of 1,700 to 2,000 fry/l for up to 30 minutes. In these tanks, liquid oxygen is bubbled through porous ceramic diffusers to maintain adequate dissolved-oxygen levels, but with no water exchange, the pH drops to 7.4 in most cases.
Pond water added to the transport tanks by small pump from the receiving ponds is used to adjust water temperature to near that of the ponds. Adjustment of pH by pumping pond water, however, is difficult, as the densely loaded fry constantly add carbon dioxide to the water through respiration. Temperature adjustment is usually achieved within 15 minutes, but pH can remain nearly unchanged.
High pH
Under these conditions, stocking of fry into ponds with pH greater than 8.0 can result in mortalities. Ponds with dense submerged weed problems – such as filamentous algae like (Chara) or (Najas) species – can be especially problematic, as they can have pH levels over 9.0 even in the early morning. Ponds freshly filled with water from the shallow alluvial aquifer were more nearly the same pH as the transport tanks.
Recommendations
Measuring pH in transport tanks and receiving ponds at the time of stocking using a pH pen is highly recommended. Transport tank water may require the addition of buffering agents if the hatchery source water is low in hardness. It is likely that most finfish species are vulnerable to pH shock if stocked seven to 14 days posthatch.
(Editor’s Note: This article was originally published in the July/August 2008 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
James A. Steeby, Ph.D.
Extension Aquaculture Specialist
National Warmwater Aquaculture Center
P.O. Box 239
Belzoni, Mississippi 39038 USA[117,100,101,46,101,116,97,116,115,115,109,46,116,120,101,64,121,98,101,101,116,115,106]
-
Charles C. Mischke, Ph.D.
Associate Research Professor
National Warmwater Aquaculture Center
Stoneville, Mississippi, USA
Tagged With
Related Posts

Aquafeeds
A look at corn distillers dried grains with solubles
Corn distillers dried grains with solubles are an economical source of energy, protein and digestible phosphorus to reduce feed costs and fishmeal usage.

Health & Welfare
A look at tilapia aquaculture in Ghana
Aquaculture in Ghana has overcome its historic fits and starts and is helping to narrow the gap between domestic seafood production and consumption. Production is based on Nile tilapia.

Intelligence
A motive, and a market, for farmed fish in Mexico
Boasting ample areas for aquaculture and a robust domestic demand for seafood – not to mention its close proximity to the U.S. market – a land of opportunity lies in Mexico. Fish farming is primed to meet its potential south of the border.

Health & Welfare
Advances in fish hatchery management
Advances in fish hatchery management – particularly in the areas of brood management and induced spawning – have helped establish aquaculture for multiple species.


