Samples analyzed by malachite green staining and nested polymerase chain reaction assay

The shrimp-farming industry faces challenging issues that include the sometimes limited availability of healthy broodstock animals and postlarvae (PL), and the emergence of various viral pathogens. Strict biosecurity measures are essential to protect the shrimp in hatcheries and ensure successful production in grow-out systems. The PL produced must of high quality and health, and free of pathogens.
Long-term study
To assist area farmers in stocking healthy fry, the authors carried out a study from November 2000 to March 2003 to assess the health of shrimp seedstock produced in hatcheries around Tamilnadu in southeastern India, and the incidence of white spot syndrome virus (WSSV) and monodon baculovirus (MBV).
During this time, 2,999 samples of black tiger shrimp (Penaeus monodon) postlarvae of various ages were received from shrimp farmers. The samples were analyzed following the method used by Systems Aquaculture Management Inc. of the Philippines, which was adopted from the SEAFDEC format.
Samples for MBV were analyzed by malachite green staining, which is part of the overall shrimp fry health analysis. Samples for WSSV were tested by the nested polymerase chain reaction (PCR) assay method.
Fry health analysis
The fry health analysis included assessment for MBV and examination of body length, rostral spine, swollen hind-gut, muscle:gut ratio, necrosis, presence of filamentous bacteria, and free and attached protozoans. Based on these parameters, the shrimp fry received an overall score indicating their health status.
An overall score greater than 93 percent, representative of predominantly healthy animals, was used as a benchmark to determine the health of different seedstock lots. MBV detection identified samples as “not satisfactory” and unfit for stocking. Other parameters like swollen hind-gut syndrome and very high necrosis also led to nonsatisfactory scores in some of the samples analyzed.
Results
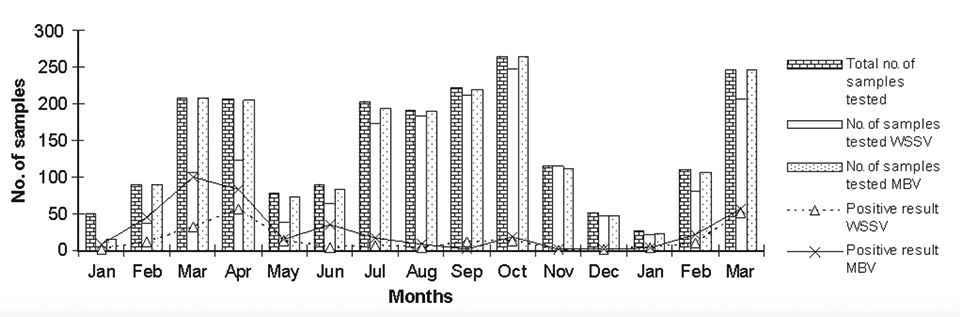
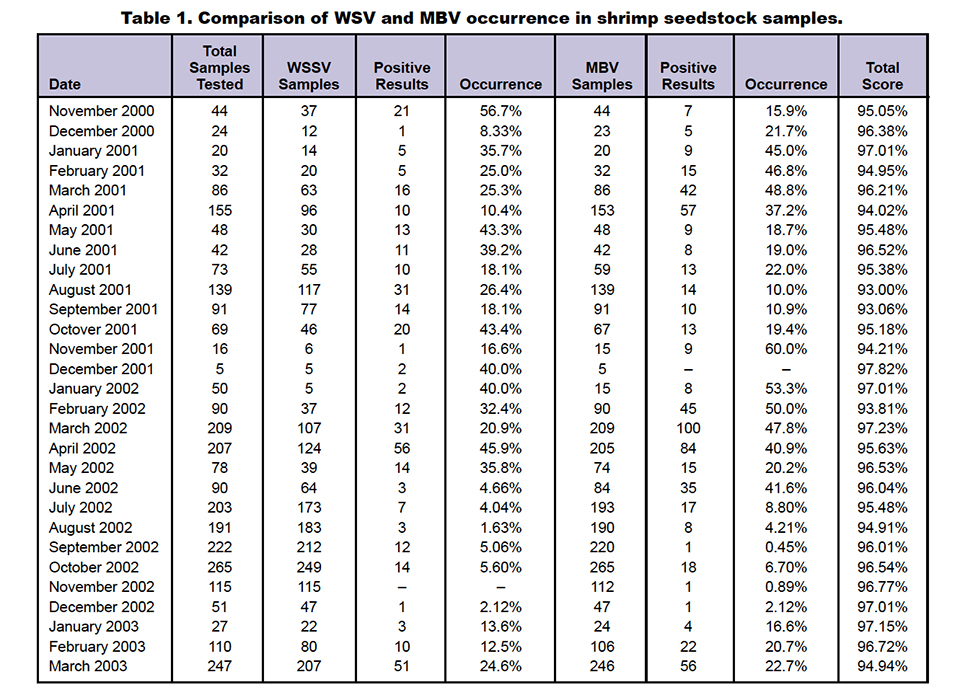
Results are presented in Table 1 and Figs. 1 and 2. The muscle:gut ratio was predominantly acceptable. Monthly averages of the samples’ health status scored 93 to 97 percent during the study period. There were only 102 samples, spread through different months, with low or poor health.
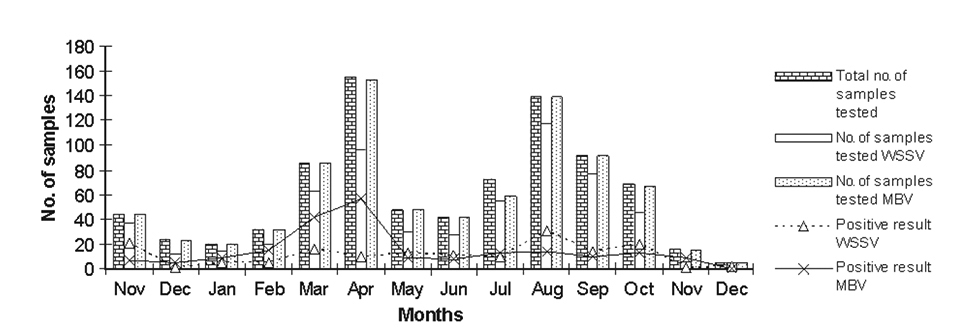
Although free and attached protozoans under acceptable levels were generally observed, no rejections were made due to high protozoan infestation. It is also interesting to note that, except for the December 2001 samples, all samples had incidences of swollen hind-gut. But only 5 percent of rejections were based on swollen hind-gut syndrome, which nevertheless affected the overall health scores. Shrimp farmers in the study area now use a health score index over 93 percent to consider a seedstock lot suitable for stocking.
MBV incidence

Of 2,904 samples tested for MBV in the study, 626 were found positive with varied degrees of infection. These were termed light, moderate, or high, based on the presence of occlusion bodies in their hepatopancreatic smears.
The least occurrences of MBV were observed during September, November, and December 2002. Although no samples tested positive for MBV in December 2001, the five samples tested that month provided inconclusive information. The maximum MBV occurrence of 60 percent was found in November 2001, but this was another month with a low number of samples.
Approximately 21.5 percent of the samples were MBV-positive during the period of study. During April 2001, 37.2 percent of 153 samples were MBV-positive, with 10 percent of 130 samples found positive in August 2001. In 2002 and 2003, the maximum number of samples tested during September and October were 220 and 265 samples, with MBV infection at 0.45 and 6.70 percent, respectively.
With 106 and 246 samples in February and March 2003, an increasing trend was observed, with 20.7 and 22.7 percent MBV positives, respectively. The lowest incidences were found in September 2002 (0.45 percent) and November 2002 (0.89 percent).
WSSV incidence
The maximum occurrence of WSSV-positive samples was recorded for November 2000, when almost 56.7 percent of the 37 samples received tested positive. In November 2001, only one of six samples received for testing was positive. Interestingly, in November 2002, all 115 samples tested were found WSSV-negative.
When representative individual fry health scores were determined to compare WSSV occurrence, 69 percent of the WSSV-positive samples had high health scores over 93 percent, and the remainder had very poor scores. It appears that even shrimp fry infected with WSSV can receive a high health score based on microscopic examination. Some WSSV-negative fry also had very poor health characteristics.
Observations
In the study, the maximum WSSV occurrence was 56.7 percent in November 2000, although WSSV was generally higher during 2001. MBV occurrence was higher during the months of January to June in both years, with 20 percent higher occurrence recorded during 2001. MBV occurrence varied 0 to 60 percent by month. The average MBV occurrence for the entire period of study was 21 percent, which was better than the results of an earlier study by the authors in the same region.
While observing the variation in MBV occurrence, an ascending trend started in January, peaked in April, and descended in May during 2001. A generally similar trend was noted in 2002, with a peak in March.
Among the fry with health scores over 93 percent, 31 percent of the samples tested positive for WSSV. Among the fry with lower scores, 20 percent were found positive. This has important implications for stocking individual seedstock lots.
Seedstock that test negative for WSSV may not be suitable for stocking if their general health score is poor, because chances are high they will become infected with WSSV after stocking. Based on these observations, it is clear that WSSV occurrence is not necessarily related to the overall health status of shrimp seedstock.
Conclusion
There is no harmonized, standard assessment available to evaluate select shrimp postlarvae. Different health standards for PL quality – from a simple observation for protozoan parasites to even Vibrio enumeration – are adopted by different diagnostic centers. Although these standards are varied, their goal is to comprehensively assess seedstock health as well as WSSV incidence.
Just checking for WSSV and stocking WSSV-negative seedstock do not guarantee healthy animals. Many farmers lose crops to WSSV in the first three weeks after stocking apparently WSSV-negative fry. This may be caused by post-larval carriers with low levels of undetected WSSV or poststocking infection due to disease vectors in the pond.
These incidents lead farmers to lose confidence in the PCR screening of fry, a generally accepted first step toward best risk management. Education is needed to help farmers understand that WSSV screening and general fry health analysis are both prerequisites for good pond yields.
(Editor’s Note: This article was originally published in the August 2004 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
S. Santhana Krishnan
Marine Technologies
56 M.G. Road
Sastri Nagar
Chennai 600 041, India
maritech@vsnl.com -
G. Sankar
Marine Technologies
56 M.G. Road
Sastri Nagar
Chennai 600 041, India
maritech@vsnl.com -
R. Nirmala
Marine Technologies
56 M.G. Road
Sastri Nagar
Chennai 600 041, India
maritech@vsnl.com -
C. Sheeba Anita
Marine Technologies
56 M.G. Road
Sastri Nagar
Chennai 600 041, India
maritech@vsnl.com -
V. Sundararaj
Marine Technologies
56 M.G. Road
Sastri Nagar
Chennai 600 041, India
maritech@vsnl.com
Tagged With
Related Posts

Health & Welfare
Development of new and real-time PCR for detection and quantification of NHPB
Authors describe conventional PCR and real-time PCR assays based on the NHPB flgE gene as alternative methods for the detection and quantification of NHPB in shrimp and shrimp-associated samples, including artemia. This newly described method will be an additional diagnostic tool for confirmation of this pathogen.

Health & Welfare
Common-sense biosecurity measures head off crop failures
Biosecurity can be defined as the operating procedures that shrimp hatchery and farm personnel use to minimize the impacts of disease on their animals. Proper biosecurity measures slow the movement of pathogens and protect unaffected areas.

Health & Welfare
Field diagnostic kits for aquaculture
New antibody-based diagnostic kits for aquaculture disease diagnosis are simple and fast, and high-sensitivity PCR testing is coming to the field, as well.

Health & Welfare
Caveat emptor when seeking for EMS solutions
The development of polymerase chain reaction testing to detect the bacteria that cause EMS is important, but confirmation by bioassay of presumptive positives to ensure pathogenicity is a prudent intermediate step.


