Vaccines, virus-free hatchery support greater production

Current aquaculture practices in developing countries are often plagued by the shortcomings of traditional aquaculture. The intensive farming patterns of backyard-style aquaculture have often caused disease outbreaks, leading to high mortality and unstable production. Attempts to normalize production have usually taken the form of antibiotic and chemical drug use, which can result in lower-quality fish products with drug residues and potential damage to the farming environment.
For example, Taiwan was previously known as the “Kingdom of Tiger Shrimp,” with an annual production of 80,000 metric tons (MT) in 1987. In 1990, the shrimp industry was nearly destroyed by white spot syndrome. When farmers changed to the culture of white shrimp, Taura syndrome appeared. Most recently, early mortality syndrome has reduced output.
The overall survival rate of fish farmed in Southern Asia is around 20 to 40 percent due to various bacterial, viral and parasitic diseases. Similar scenarios have occurred globally.
To meet the increase in global demand for seafood, the aquaculture industry clearly needs new concepts, products and biosecurity technology to address these significant bottlenecks.
Vaccines
As vaccines have been employed successfully to control diseases in human and domestic land animals, such prophylactic approaches have prevented disease outbreaks, decreased the abuse of antibiotics and assisted the industrialization of cold-water fish aquaculture in developed countries. Vaccines have significantly advanced the salmon industry in Norway and the sea bream and sea bass industries in the Mediterranean Sea.
Unlike land animals that live in air and have constant body temperatures, fish live in water where the temperature, salinity and farming methods vary and alter the population of microorganisms. Warmwater fish, in particular, have diverse epidemics and pathogens in different farming environments. Therefore, there is need for the development of fish vaccines using concepts appropriate for aquaculture in developing countries, where 80 percent of fish farming takes place.
Grouper aquaculture
Grouper are high-value fish well-liked by Asians and Arabians in the Middle East. They are familiar to consumers and have a stable market demand, which makes the species a candidate for expanded culture.
Grouper are demersal fish that live in coral reefs and are difficult to catch by conventional net fishing, so illegal methods of grouper fishing are used, including explosives and poisons that have destroyed their natural habitats. Consequently, several species of grouper have been listed as near endangered and even endangered, so farming of grouper is necessary to supply the market demand and reduce fishing pressures.
According to Food and Agriculture Organization of the United Nations estimates, in 2020, the grouper demand will reach 500,000 MT – met in equal shares by fishing and aquaculture. Grouper farming will expand threefold from 2008 levels, with accompanying needs for additional grouper fry and culturing technology. Most grouper are currently farmed in Asia. Among the top producing countries are China (61 percent), Taiwan (14 percent) and Indonesia (13 percent), followed by Malaysia (9 percent).
However, in Taiwan and neighboring south Asian countries, grouper fingerling production has suffered due to major mortalities from severe nervous necrosis virus (NNV) infection that resulted from careless farming behavior. At the grow-out stage, grouper have been found to be infected by NNV, Irido virus and bacterial pathogens such as Vibrio species, Aeromonas, Streptococcus and parasites.
Vaccine development
In 2001, the authors built a marine fish research team at National Cheng Kung University in Taiwan that has since grown into a team of five principal investigators and over 40 graduate students. The group selected grouper as a target fish for which to develop new and sustainable farming technology. In the process, the authors developed an oral vaccine for grouper to control NNV infection at the larval stage and multivalent injective bacterial and viral vaccines for the grow-out stage.
As NNV is mostly found at the juvenile larval and postlarval stages, vaccines must be administered early before infection. While several NNV subunit vaccines have been developed, there are issues regarding administering those vaccines to larvae while they are sensitive to handling stress. Injective and immersive immunizations are impractical, leaving oral vaccination the only option.
The primary bottlenecks for oral vaccination are palatability and the gastrointestinal digestion of the antigen. As a result, the success of an oral vaccine depends on its ability to attract the fish to eat the vaccine, protecting the antigen during digestion and effectively delivering it to the hindgut of the fish, which is near the immune organ.
The authors have developed an oral vaccine using artemia or rotifers to encapsulate the inactivated recombinant subunit vaccine containing the specific antigen. In addition, the authors established a multivalent injective vaccine that with one immunization can prevent most viral and bacterial diseases for the grow-out stage.
NNV-free hatchery
NNV has been found in the waters of most farming areas, as well as in broodstock, fertilized eggs and commercial live starting feeds at current outdoor grouper hatcheries. To help control further viral epidemics, the authors designed and constructed a pilot-scale virus-free grouper hatchery.
This facility and its stable supply of virus-free larvae enabled detailed studies on the parameters of grouper embryo development, which included step-by-step analyses of their chemical, physical and nutritional requirements. These data base then facilitated the assembly of electronic standard operating procedures for the indoor production of disease-free fingerlings.
In the virus-free hatchery, over 60 consecutive successful fingerling production cycles were achieved. That demonstrated the ability of the indoor hatchery to produce healthy NNV specific pathogen-free (NNV-SPF) grouper fingerlings (Table 1).
Yang, Performance of grouper fingerlings, Table 1
| Method | Reproduction Frequency | Deformity Rate (%) | Productivity (fish/mt) | Subsequent Survival Rate |
|---|---|---|---|---|
| Outdoor hatchery | 1/10 | 20-40% | 2-30 | 20-50% |
| Indoor hatchery | Stable | Under 5% | Over 1,000 | 80% |
Vaccinated fingerlings
After the injective vaccine was developed, it was very difficult to get farmers to vaccinate their fish. Immunization by injection of 5-cm-long fingerlings is labor intensive and highly technical. It was also difficult to convince farmers to inject their healthy fish for preventive purposes.
Therefore, the NNV-SPF fingerlings were vaccinated before shipping to farmers. The immunized fingerlings are resistant to several diseases and can elevate survival rates and decrease production costs (Fig. 1).
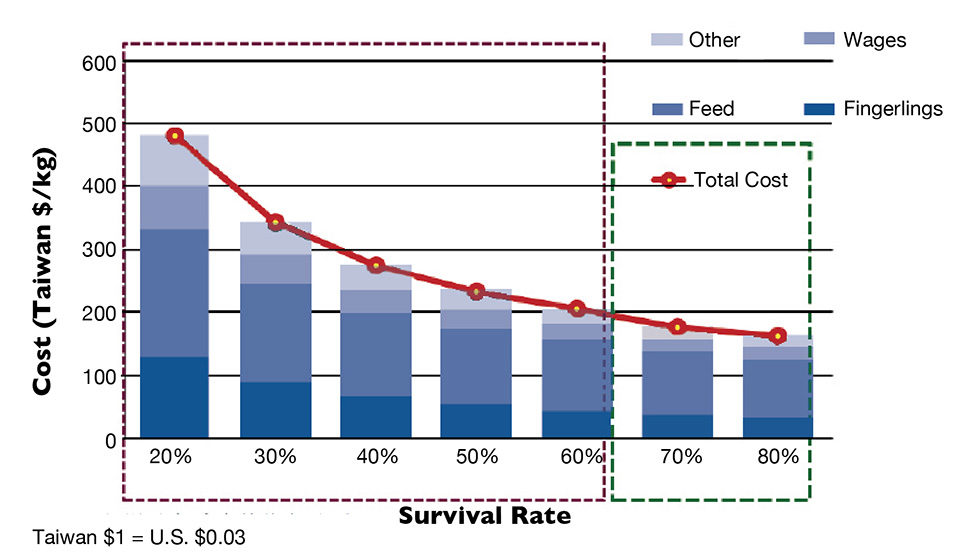
The performance of a million fingerlings was tracked at over 30 farms in various locations in Taiwan with different farming conditions and stocking densities. In a one-year grow-out period, over 80 percent of the farms achieved 75 to 85 percent survival rates, versus the 20 to 40 percent survival typically noted using other fingerlings.
Market potential: frozen fillets grouper are mainly produced to supply the whole fish market, especially the live fish market for Chinese restaurants. The distribution of live whole fish requires sophisticated and expensive transporting methods. The live fish form also limits distribution to other markets. Production of frozen fish fillets would allow the distribution of grouper to international market outlets.
Several species of grouper have been successfully reared using the indoor hatchery, including Epinephelus maculates, E. fuscoguttatus, E. coioides, E. lanceolatus, Plectropomus leopardus, the coldwater E. bruneus and the hybrid Sabah giant grouper. Giant grouper is a very high-quality white-meat fish that grows quickly – reaching 15 to 25 kg in three years versus the growth of salmon to 4 to 8 kg in the same period. As a demersal fish, grouper should also have better feed conversion.
Editor’s Note: This article is based on the authors’ research to develop vaccines to protect cultured grouper from various diseases. This work was considered for the Novus Global Aquaculture Innovation Award, the first of which was presented by the Global Aquaculture Alliance at the GOAL 2013 conference in Paris, France.
(Editor’s Note: This article was originally published in the January/February 2014 print edition of the Global Aquaculture Advocate.)
Authors
-
Huey-Lang Yang, Ph.D.
Merit Ocean Biotech
Number 51, Keji 5th Road
Annan District
Tainan City 709 Taiwan[109,111,99,46,108,105,97,109,103,64,103,110,97,108,121,101,117,104,103,110,97,121]
-
Han-You Lin, Ph.D.
Institute of Biotechnology
National Cheng Kung University
Tainan, Taiwan -
Chi-Chu Lin
Institute of Biotechnology
National Cheng Kung University
Tainan, Taiwan
Tagged With
Related Posts

Intelligence
Bahamas venture focuses on grouper, other high-value marine fish
A new venture under development in the Bahamas will capitalize on Tropic Seafood’s established logistics and infrastructure to diversify its operations from processing and selling wild fisheries products to include the culture of grouper and other marine fish.

Aquafeeds
Increased density improves grouper feeding response, growth
A growth trial using hatchery-reared grouper was carried out to study the effects of stocking density on feed intake and subsequent growth. Contrary to common perceptions, fish stocked at the highest density had higher feed intake and body weights.

Aquafeeds
Soy trials replace fishmeal in grouper feeds
Although groupers present desirable traits as an aquaculture species, the high cost of feed for the carnivorous fish limits expansion of grouper culture. The author performed two trials to evaluate the performance of tiger grouper juveniles fed soybean meal-based feeds.

Health & Welfare
A look at tilapia aquaculture in Ghana
Aquaculture in Ghana has overcome its historic fits and starts and is helping to narrow the gap between domestic seafood production and consumption. Production is based on Nile tilapia.


