Vaccination effective against main strep species

As tilapia become increasingly popular as a farmed fish, the international trade in tilapia is growing rapidly. Relatively easy to breed, they grow well in a variety of habitats and provide year-round yields.
However, with the increasing popularity and intensification of tilapia culture, it has become apparent that these fish are susceptible to a variety of infectious diseases with bacterial or viral etiology and potentially devastating consequences for farmers. Foremost among these is streptococcosis, a septicemic disease responsible for significant mortality in farmed tilapia.
Main strep species
Two species of streptococci, Streptococcus agalactiae and S. iniae, are generally considered the most important agents of streptococcal disease in tilapia. Tilapia infected with S. agalactiae or S. iniae exhibit similar clinical signs, including lethargy, abnormal swimming, C-shaped body posturing, unilateral or bilateral exophthalmia and lack of appetite.
These similarities make on-farm diagnosis difficult and necessitate the analysis of bacterial isolates by conventional phenotypic methods or polymerase chain reaction using species-specific oligonucleotide primers.
Prevalence
To better understand the importance of these two pathogens to the tilapia industry, the authors have during the last nine years identified over 1,000 bacterial isolates from tilapia reared at 74 sites in 14 countries. In common with other investigators, they found that streptococcal species account for more than half of all bacteria identified.
Interestingly, while S. iniae is the most commonly reported streptococcal pathogen of fish, the authors’ data showed that S. agalactiae is more prevalent in tilapia. While the reason for this apparent predominance is not known, a recent comparative histopathology study of S. iniae– and S. agalactiae-infected tilapia revealed subtle differences in the pathogenesis of the two bacteria, suggesting that tilapia may control natural infections with S. iniae more effectively than those caused by S. agalactiae.
S. agalactiae is the only streptococcal species that carries the group B antigen and as such is often referred to as simply group B streptococcus. Although more commonly associated with disease in human or bovine hosts, where it causes neonatal sepsis and mastitis, respectively, fish-pathogenic S. agalactiae was documented as early as 1966, when a non-hemolytic group B streptococcus was identified as the cause of two epizootics in golden shiners.
In recent years, there has been a significant increase in the number of published investigations on S. agalactiae disease in tilapia. In 1994, Avi Eldar and colleagues identified a new serologically untypeable streptococcal pathogen of tilapia for which they proposed the name S. difficile. The species name was later corrected to S. difficilis, but subsequent studies demonstrated that S. difficilis is a variant non-hemolytic group B streptococcus that is genetically identical to S. agalactiae.
Biotypes
Detailed analysis of S. agalactiae isolates suggested the presence of two distinct clusters or biotypes that differ in a variety of biochemical and phenotypic characteristics. Strains of biotype 1 are typically beta-hemolytic, ferment a variety of sugars including trehalose and galactose, and grow well at 37 degrees-C. In contrast, all the strains of biotype 2 tested by the authors were non-hemolytic, more restricted in their utilization of sugars and grew poorly, if at all, at 37 degrees-C. The phenotypic characteristics of the biotype 2 strains closely resembled those described previously for S. difficilis.
In the authors’ experience, the two S. agalactiae biotypes cause subtly distinct disease syndromes. Biotype 1 tends to infect fish throughout the production cycle from juvenile to growout, while biotype 2 causes disease predominantly in larger fish.
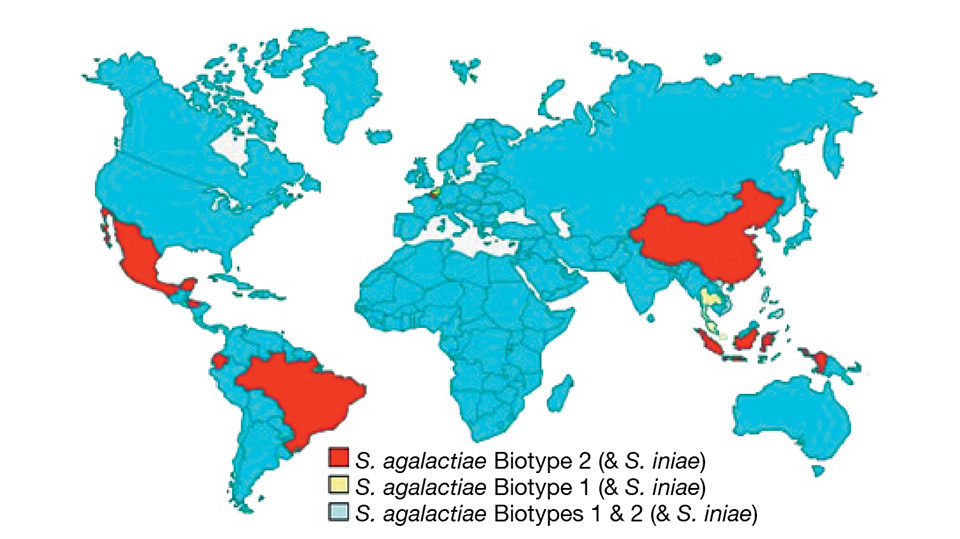
Analysis suggested that the S. agalactiae biotypes are present in distinct geographical zones. From the authors’ worldwide sampling, S. agalactiae biotype 2 is the most prevalent and geographically diverse of the streptococcal pathogens. S. agalactiae biotype 2 is found in Asia (China, Indonesia, Vietnam and the Philippines) and Latin America (Ecuador, Honduras, Mexico and Brazil).
In contrast, S. agalactiae biotype 1 is the dominant streptococcal pathogen for tilapia in Thailand, Malaysia and Singapore. S. iniae is often identified in association with S. agalactiae biotypes 1 or 2 in China, Ecuador, Honduras, Indonesia, the Philippines and Thailand. Only in the Philippines and Vietnam are S. agalactiae biotype 1, S. agalactiae biotype 2 and S. iniae found in the same country.
There are no obvious geographical, physiological or environmental explanations for the country-specific distribution of S. agalactiae biotypes 1 and 2. As such, it is prudent to consider the possibility that this will break down over time, probably through trade in live fish.
Vaccines
Vaccines to protect against S. agalactiae infection in tilapia have been described by various authors. However, it is difficult to conclude from the studies if the vaccine and challenge strains were from the same or different biotypes.
To determine if their classification of the fish-pathogenic S. agalactiae has consequences for the development of vaccines to control streptococcosis, the authors assessed in a laboratory challenge the ability of biotype-specific vaccines to protect against lethal challenge with S. agalactiae biotype 1 or 2 strains.
Vaccinated and unvaccinated control tilapia were challenged at three weeks post-vaccination. Fish were observed for 15 days after challenge, and mortality was recorded daily. Post-mortem recovery of the challenge organism was performed on fish that died during the observation period and on all surviving fish at the end of the observation period.
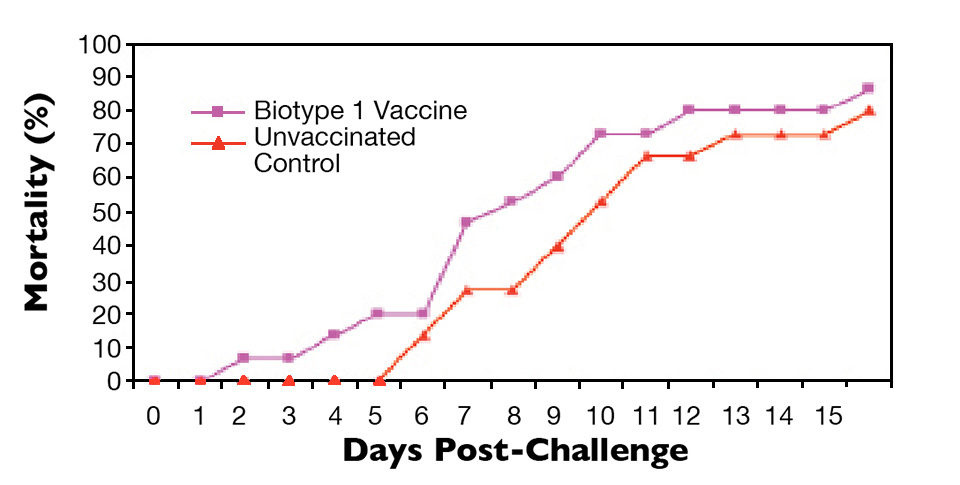
The results of representative experiments are shown in Figs. 1 to 4. Tilapia vaccinated with an experimental S. agalactiae biotype 1 vaccine were protected against lethal challenge with a virulent S. agalactiae biotype 1 strain (Fig. 1). However, no protection against challenge with a virulent biotype 2 strain was observed in the biotype 1-vaccinated fish (Fig. 2).

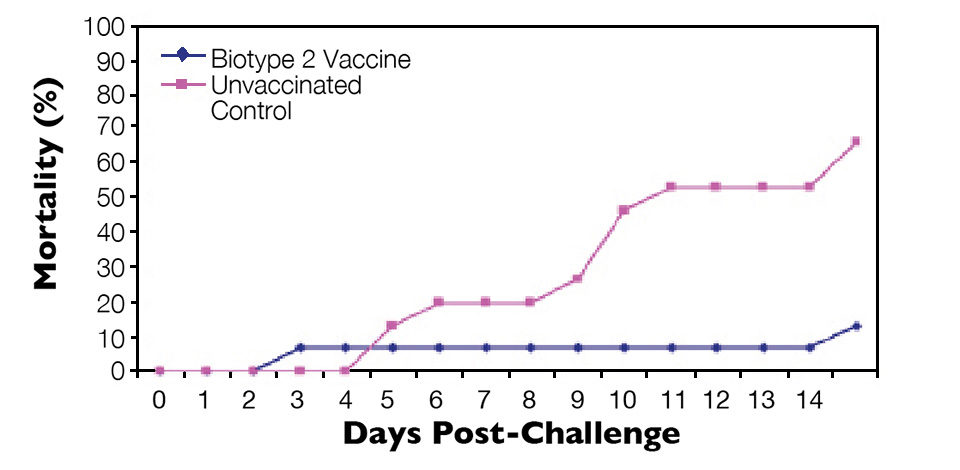
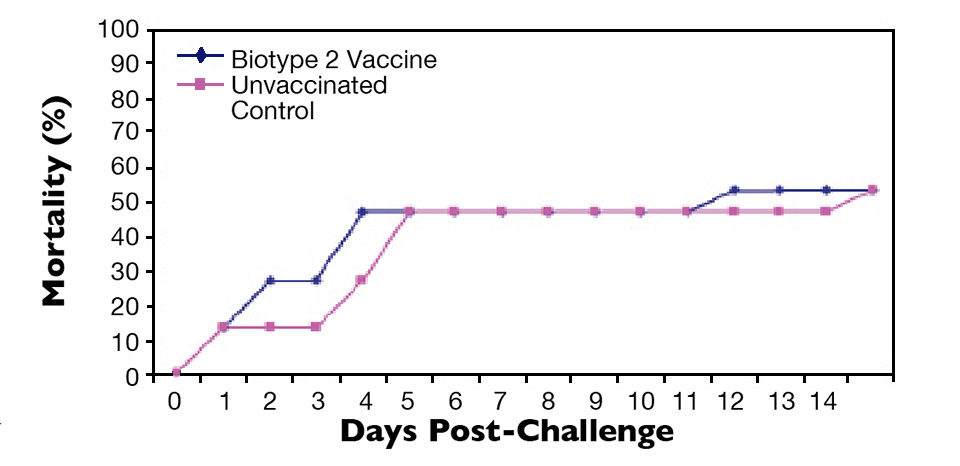
Similarly, fish vaccinated with an S. agalactiae biotype 2 vaccine were protected against lethal S. agalactiae biotype 2 challenge (Figure 3) but not challenge with a virulent biotype 1 strain (Figure 4). Thus, vaccination with biotype-specific bacterin vaccines induced biotype-specific protection against mortality caused by S. agalactiae.
For any tilapia operations suffering from a streptococcal disease problem and considering the use of vaccination as a means of control, it is vitally important that they determine which streptococcal species or biotype is causing their problem so that the most appropriate vaccine can be chosen.
(Editor’s Note: This article was originally published in the July/August 2009 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
Brian Sheehan, Ph.D.
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847[109,111,99,46,116,101,118,114,101,116,110,105,46,112,115,64,110,97,104,101,101,104,115,46,110,97,105,114,98]
-
L. Labrie, DVM
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847 -
Y.S. Lee, B.S.
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847 -
F.S. Wong, B.S.
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847 -
J. Chan, B.S.
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847 -
C. Komar, DVM
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847 -
N. Wendover, B.S.
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847 -
L. Grisez, Ph.D.
Intervet/Schering-Plough Animal Health
1 Perahu Road
Singapore 718847
Tagged With
Related Posts

Health & Welfare
Aquaculture disease experts to download at GOAL 2016
At this year’s GOAL conference in Guangzhou, China, leading animal welfare and aquatic animal disease experts will share knowledge about how producers can gird their operations. EMS, EHP, streptococcosis and sea lice will be addressed, as will the latest in area management.
Health & Welfare
Developing live bacterial vaccines by selecting resistance to antibacterials
As wide use of antibiotics has led to antibiotic resistance in fish pathogens, vaccines present an alternative control method to prevent bacterial diseases.

Health & Welfare
Parasite treatment reduces F. columnare infection in tilapia
The authors conducted a study to evaluate whether treatment of Trichodina-parasitized tilapia with formalin would improve fish survival and reduce F. columnare infection. Tilapia not treated with formalin showed significantly higher mortality than treated fish.

Health & Welfare
Beneficial microbes and pathogen control
A range of alternative products is available to improve animal health and water quality, and control pathogen loads. Beneficial microbes produce antimicrobial compounds and suppress pathogen proliferation.


