Low temperatures not conducive to fast growth, but water quality yields excellent product

Scallop culture is a well-established industry in some parts of the world. Japan began investigating the potential for scallop culture in the 1930s and started commercial production in the mid-1960s. Building on Japan’s success and borrowing from the procedures developed there, China and Chile began their own scallop culture industries in the 1980s.
Additional small but developing culture industries exist in eastern Canada, Russia, Great Britain, France, Norway, Ireland, Italy and Spain. In spite of the success of scallop culture elsewhere in the world, efforts in the United States fail to meet domestic demand and remain limited to small-scale operations.
Purple-hinge rock scallops
In the waters of the U.S. state of Alaska, the Pacific weathervane scallop, Patinopecten caurinus, is the only commercially harvested scallop species. The purple-hinge rock scallop, Crassadoma gigantean, has potential for aquaculture, and is highly prized by local communities and harvested for subsistence in coastal Alaska.
Purple-hinge rock scallops are found along the U.S. Pacific coast from Baja, California, northward to Alaska, although their distribution is patchy throughout the range. Unlike their free-swimming pectinid relatives, the scallops have developed an affinity for permanent attachment. in rocky substrates. This presents challenges for commercial harvest, but is a potential advantage for mariculture.
Aquaculture of purple-hinge rock scallops has been investigated in California, and while results were promising, there were barriers to implementing commercial culture operations. Research conducted in 1989 by Neil Bourne in British Columbia showed promise for mariculture in a more northerly location. Renewed interest in Alaska resulted in a study by the authors that further determined the feasibility of culturing this species.
Scallop projects
A survey of Alaska’s shellfish-farming industry in 1997 recognized the importance of species diversity, and the production of high-value shellfish became a top priority. In January 2006, an aquaculture planning session sponsored by the governor’s office again supported the development of new species as a priority.
In response, the Alaska Sea Grant Marine Program, Qutekcak Shellfish Hatchery and Alaskan Shellfish Growers Association invested in research on hatchery production and field growout trials to develop farming practices for the purple-hinge rock scallop. Native to Alaskan waters, purple-hinge rock scallops are biologically suitable for aquaculture and sell for over U.S. $3/scallop on the local market.
Hatchery production research started in 1997 with funding from the National Marine Fisheries Services. Over a two-year period, multiple reproduction and larval-rearing trials were performed, with the first successes in 1999. In the following year, a sizable population produced enough seed to begin field growout trials. The most significant challenge to the hatchery was low survival from the pedivelliger stage to a seed size of 7.5 mm.

Grow-out work
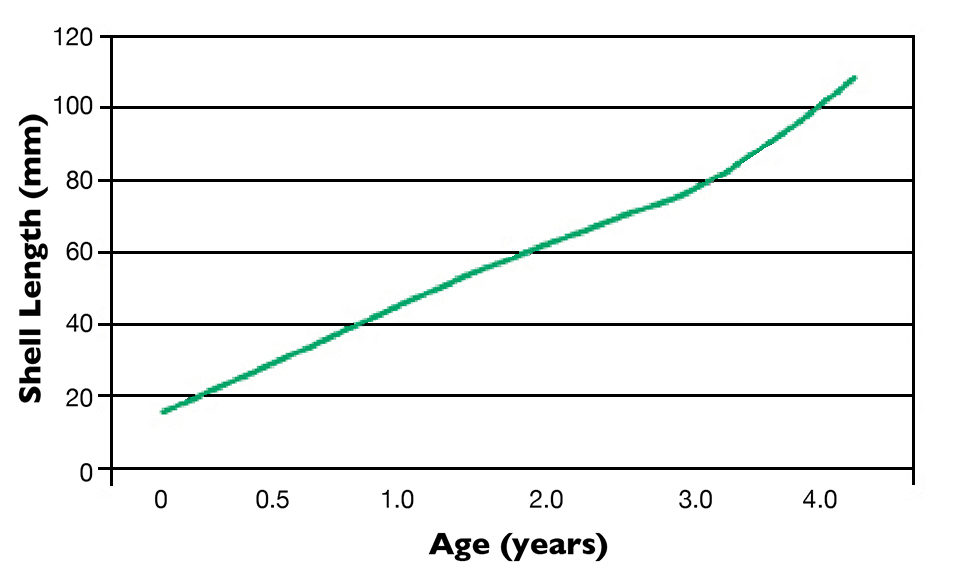
The Alaska Science and Technology Foundation has funded subsequent field grow-out research since 2001, with purple-hinge rock scallop seed 31.1 mm in shell length delivered to the Pearl of Alaska farm near Kake in southeastern Alaska. The scallops were nursery cultured in lantern nets, where they reached 52 mm in length. Survival during this initial phase was density dependent, varying from 17 percent at a density of 300 scallops/chamber to 50 percent when 100 scallops were stocked per chamber. For grow-out, the scallops were moved to nornets and raised to 4.3 years of age. A nornet is a production unit composed of a “stack” of rigid perforated disks separated by about 10 cm and encased in netting. Scallop seeds are placed on each disk.
Nornets were used because as scallops grow, they incorporate the substrate into their bottom shells. With lantern nets, the netting would be grown into the shells. Removing scallops from the net would require destroying the nets. The nornets’ chamber flooring of thick, perforated plastic prevents scallops from growing into the webbing, simplifying sampling.
Measurements of the scallops’ adductor muscles were taken at ages 2 and 4 years (Table 1). Shell growth for the four-year period is shown in Fig. 1.
RaLonde, Purple-hinge rock scallop, Table 1
| Diameter (mm) | Height (mm) | Weight (g) | Recovery (%) | No./ lb | |
|---|---|---|---|---|---|
| Age 2 | 27.59 ± 1.25 | 11.87 ± 0.93 | 7.89 ± 0.82 | 10.41 ± 0.56 | 62.66 ± 7.17 |
| Age 4 | 32.34 ± 1.37 | 20.53 ± 1.26 | 19.09 ± 1.87 | 14.23 ± 0.01 | 26.94 ± 2.90 |
Scallop traits
While purple-hinge rock scallops are locally prized, they would be a new species for the seafood market – where quality assessment is necessary to promote acceptance. Ten adult purple-hinge rock scallops were harvested from pens at Elfin Cove Oysters in Port Althorp on the north end of Chichagof Island in southeast Alaska and shipped live to the Fishery Industrial Technology Center in Kodiak. The scallops were held at 4 degrees C, and processing was carried out within two days of arrival.
The wet weights of whole scallops, shells, gonadal-visceral (body) tissues and adductor muscles were measured using an electronic digital scale. Shell length, width and depth, and adductor muscle length and width were determined using a cloth tape measure and electronic digital calipers.
The scallops were shucked after whole weights were recorded. The adductor muscles were individually vacuum packed and stored at minus-30 degrees-C until analysis. A muscle condition index (MCI) assessing the physiological condition of the adductor muscle relative to total soft tissue was calculated for the samples according to the formula MCI ( percent) = weight of adductor muscle/weight of total soft tissues x 100.
The purple-hinge rock scallop is a large species with massive shells and large adductor muscles (Table 2). The range of the 10 specimen whole weights was 561 to 1,202 grams. Shell heights ranged from 138 to 189 mm. Yields of edible adductor muscle were less than 10 percent. However, MCI values were between 36.0 and 50.9.
RaLonde, Morphological measurements, Table 2
| Whole Weight (g) | Adductor Muscle Weight (g) | Adductor Muscle Yield (%) | Shell Height (mm) | Muscle Condition Index | |
|---|---|---|---|---|---|
| Average | 820.0 ± 200.1 | 52.1 ± 21.0 | 6.3 ± 1.6 | 167.2 ± 13.5 | 44.2 ± 4.4 |
The results reflected observations made by other researchers on the size and condition of purple-hinge rock scallops. Individuals can reach shell heights of 250 mm, and the shells become massive and heavy. David Leighton reported purple-hinge rock scallop adductor muscle weights from 50 to 80 grams, and muscles at 30 to 55 percent of soft body weight, reported in this study as MCI. In comparison, weathervane scallops, which can also reach shell heights of 250 mm, typically have adductor muscles in the 27- to 48-gram range and yields of 10 to 12 percent.
Composition
The adductor muscle and digestive gland are the primary storage sites of nutrient reserves in scallops. Carbohydrate, in the form of glycogen, and protein are stored in the adductor muscle, while lipid is stored in the digestive gland. The gross composition of rock scallops is shown in Table 3.
RaLonde, Gross proximate composition, Table 3
| Moisture (%) | Protein (%) | Glycogen (% | Ash (%) | Lipid (%) | |
|---|---|---|---|---|---|
| Average | 76.5 ± 0.7 | 19.7 ± 1.6 | 1.4 ± 0.4 | 1.6 ± 0.8 | 0.8 ± 0.2 |
Purple-hinge scallop adductor muscle is lean due to its low lipid content. Lipids in the muscle are mainly composed of phospholipids. Furthermore, purple-hinge scallop adductor muscle is an excellent source of high-quality marine protein. With regards to flavor, glycogen, together with a variety of amino acids, impart desirable sweetness to the product.
Perspectives
The purple-hinge scallop is a specialty seafood item that provides an opportunity for shellfish growers in Alaska to diversify their farm production. The scallops can easily fit in with suspended oyster farming.
The low water temperatures in Alaska are not conducive to fast growth of shellfish, but the pristine water quality yields excellent product quality. Because of scallops’ long grow-out period to market size, research continues to develop grow-out techniques that require minimal labor for farmers.
(Editor’s Note: This article was originally published in the May/June 2012 print edition of the Global Aquaculture Advocate.)
Authors
-
R. RaLonde
Alaska Sea Grant Marine Advisory Program
University of Alaska – Anchorage
1007 West 3rd Avenue, Suite 100
Anchorage, Alaska 99501 USA[117,100,101,46,97,107,115,97,108,97,64,97,114,105,101,118,105,108,111,99,97]
-
K. Brenner
Fishery Industrial Technology Center
University of Alaska – Fairbanks,
Kodiak, Alaska, USA -
A.C.M. Oliveira
Fishery Industrial Technology Center
University of Alaska – Fairbanks,
Kodiak, Alaska, USA
Tagged With
Related Posts

Intelligence
Aquaculture UK: Stepping up to the plate
There’s considerable opportunity to grow the UK aquaculture industry. At the Aquaculture UK exhibition and conference in Aviemore, Scotland shows the way.

Innovation & Investment
Aquaculture is winning, Rabobank analyst explains
Aquaculture is the “winning protein,” according to a new Rabobank report that its author, Gorjan Nikolik, said is intended to draw the bank’s agro-industry clients to opportunities in the fish farming business.

Innovation & Investment
Aquaculture planning, development in Brazilian federal waters
The aquaculture industry in Brazil is moving toward further expansion with the support of the federal government. A key strategy of the More Fishing and Aquaculture plan is the development of aquaculture in federal waters. The plan promotes sustainable development of fisheries and aquaculture by linking those involved and consolidating state policies addressing social inclusion, security and food sovereignty. Tilapia is the main farmed fish, although tambaqui and others have potential for large-scale production due to their wide acceptance by consumers.

Intelligence
Behold the nutritious oyster
Oysters provide important, natural filtration of water and are an important component of many healthy coastal ecosystems because their active filtering can help improve and maintain water quality. For many coastal communities, oysters are an important food resource and excellent sources of protein and amino acids, zinc, selenium, iron and B-vitamins.


