Ciliated parasite can lead to white spot disease

White spot disease is a parasitic disease commonly seen worldwide in freshwater fish. Infected fish show small white spots on the skin, fins and gills. When conditions are favorable for the disease, entire fish populations can be lost within a few days.
White spot disease is caused by the ciliated parasite Ichthyophthirius multifiliis, commonly referred as Ich. It has three stages in its life cycle: a reproductive tomont, an infective theront and a parasitic trophont. The Ich infection is difficult to treat by chemotherapy because the parasite penetrates fish skin and gills.
Intensive aquaculture requires methods to prevent and control white spot disease to minimize losses. Vaccination against the parasite is an alternative to chemical treatment, as fish immunized with Ich antigens acquire immunity against the parasite.
Immunization trial
The authors conducted a trial using channel catfish to evaluate the effects of Ich antigens and routes of immunization on immune protection in fish. Catfish of 16-gram average weight were divided into six groups of 28 fish each and immunized as follows:
- Immersion at a dose of 20,000 live theronts per fish, followed by daily formalin treatment for five days
- Intraperitoneal (I.P.) injection at a dose of 20,000 live theronts per fish
- Immersion at a dose of 100 sonicated trophonts per gram of fish
- I.P. injection at a dose of 20 sonicated trophonts per gram of fish
- I.P. injection with 5 percent bovine serum albumin (BSA)
- Non-immunized control.
Four fish were sampled from each immunized group 12 and 21 days after immunization to determine serum antibody levels against Ich. Then all fish were challenged by exposure to Ich infective theronts at a dose of 15,000 theronts/fish to determine fish infection level and survival.
The catfish showed immune responses in three groups: those immunized with live theronts by immersion, those that received live theronts by I.P. injection and fish injected with sonicated trophonts. The immunized fish demonstrated high anti-Ich antibody levels 12 days after immunization, as measured by Ich immobilization assay. The assay is a serological method that measures the amount of antibodies to Ich in blood serum.
For the three groups, serum antibodies against Ich were 10 to 30 times higher than in control fish. No antibody was detected in the other test groups.
All catfish immunized with live theronts by immersion or I.P. injection and 90 percent of the catfish immunized by injection with sonicated trophonts survived theront challenge. Fish that received the other forms of immunization died within two weeks after parasite challenge.
Trophont immunization trial
Fish immunized with live theronts by either immersion or injection were protected. However, fish immunized with inactivated trophonts showed various results in other studies.
To evaluate the effects of inactivated trophonts on immune response, a trial was conducted using 250 channel catfish. Trophonts were collected from Ich-infected fish and inactivated with formalin or freezing.
The fish were immunized intraperitoneally with 1 percent formalin-inactivated trophonts, 3 percent formalin-inactivated trophonts or freeze-thawed trophonts at a dose of 20 trophonts per gram of fish. Positive and negative control catfish were immunized by I.P. injection with 20,000 live theronts per fish and 5 percent BSA, respectively. A blood sample was taken from six fish from each immunized group at 14, 28 and 50 days following immunization to determine antibody levels.
Antibody levels in the immunized fish were six to eight times higher than in the BSA control fish. The survival rates in fish immunized with inactivated trophonts were 33.3 to 44.3 percent – significantly higher than for the control fish, which had zero survival.
Trophont dose trial
In a third study, channel catfish about 12 g in weight were divided into four groups to evaluate the effects of trophont antigen dose on protective immunity. The fish were immunized with formalin-inactivated sonicated trophonts by I.P. injection at doses of 5, 10 or 20 trophonts per gram of fish. Control catfish were injected with 5 percent BSA.
Four fish from each group were sampled at days 14 and 22 post-immunization, and serum antibody levels were determined. Thirty fish per group were challenged on day 23 by exposure to Ich theronts at the dose of 15,000 theronts and mortality was observed for three weeks.
The fish immunized with trophonts at 10 or 20 trophonts per gram of fish showed four to nine times higher antibody levels than fish immunized at 5 trophonts per gram and 20 to 40 times higher levels than in the control fish. Fish immunized with 10 or 20 trophonts per gram of fish showed less than 50 established trophonts per fish compared to levels of 100 trophonts per fish for the BSA control. Fish immunized with the higher doses of trophonts had 60 to 63 percent survival, while those receiving 5 trophonts per gram fish had 23 percent survival, and the controls with no trophont had less than 7 percent survival.
Live theronts, inactivated trophonts
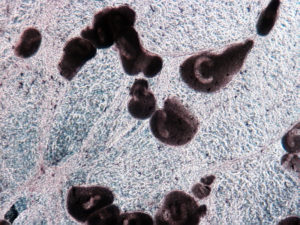
Live theronts stimulate fish to develop a strong immune response and provide good protection. Immunization with live theronts by I.P. injection is generally safe to fish and does not cause infection. Injection, however, is laborious and impractical for most cultured fish. In addition, live theront exposure following treatment is a controlled infection caused by a virulent parasite. The application is usually done only in laboratory trials, and cautions need to be taken to prevent fish infection or mortality.
The methods used to administer inactivated trophonts influence immune protection. Sonicated trophonts administered by immersion did not induce protective immunity due to the large size of the parasite protein antigen, which cannot get through fish skin. When administered by I.P. injection, the dosages of trophonts used to immunize the fish influence immune protection. Fish were protected when they were immunized with at least 10 trophonts per gram of fish.
One major obstacle for the development of vaccines against Ich is the lack of antigen sources. There is no method available for large-scale culture of the parasite. Further research is needed to overcome this serious limitation by developing feasible culture methods for the parasite or using molecular techniques to produce large quantities of recombinant protective antigens.
(Editor’s Note: This article was originally published in the July/August 2009 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-
De-Hai Xu, Ph.D.
United States Department of Agriculture
Agricultural Research Service
Aquatic Animal Health Research Laboratory
990 Wire Road
Auburn, Alabama 36832 USA[118,111,103,46,97,100,115,117,46,115,114,97,64,117,120,46,105,97,104,101,100]
-
Phillip Klesius, Ph.D.
United States Department of Agriculture
Agricultural Research Service
Aquatic Animal Health Research Laboratory
990 Wire Road
Auburn, Alabama 36832 USA -
Craig Shoemaker, Ph.D.
United States Department of Agriculture
Agricultural Research Service
Aquatic Animal Health Research Laboratory
990 Wire Road
Auburn, Alabama 36832 USA
Tagged With
Related Posts

Health & Welfare
Candidate gene markers for selective breeding of CyHV-3-resistant carp, koi
Common carp and koi producers around the world have suffered substantial financial losses due to a contagious disease caused by cyprinid herpesvirus-3.
Health & Welfare
ELISA kits offer quantitative analysis of trifluralin in fish
Antibody-based enzyme-linked immunosorbent assay (ELISA) tests are proven, sensitive, high-throughput alternatives to more costly and complex test methods for the detection of herbicide residues and other chemicals.

Health & Welfare
Oral vaccine delivery effective in reducing salmon diseases
Oral vaccination can be done with virtually all fish sizes and is a relatively stress-free and inexpensive approach. The authors developed a novel oral vaccine delivery system that was found effective against infectious salmon anemia and salmonid rickettsial septicemia at salmon farms in Chile.

Intelligence
A brief look at genetically modified salmon
If approved by FDA, fast-growing genetically modified salmon will provide a safe and nutritious product similar to other farmed Atlantic salmon.



