A look at nutritional contributions and shrimp production

Because closed aquaculture systems have very low water exchange and controlled inputs, and because they typically have a smaller footprint than traditional open ponds, these systems are receiving increased attention to enhance biosecurity and minimize water use to culture marine species inland.
These intensive, closed systems can be established indoors and near major consumer centers for year-round production, and are gaining popularity in some regions, including the United States, where indoor shrimp farming has an interesting opportunity to develop further.
Clear-water recirculating aquaculture systems (CW) and biofloc (BF) technology systems are two categories of closed aquaculture systems. CW systems usually involve an external biofilter for nitrifying bacteria and filters for solids removal from the water. Some systems also have UV lamps for water sterilization. These systems typically have more filtration components and higher capital and operational costs vs. BF systems. But by setting up biofiltration externally with consistent conditions, CW systems may provide more system control and stability, especially regarding nitrogen cycling.
BF systems have a significant amount of particulates and a dense microbial community, and their only external filtration is usually a solids filter to manage particulates. Although these systems involve less equipment, usually have lower capital costs, and biofloc particles may provide supplemental nutrition for shrimp, BF systems are generally more difficult to manage and require more aeration equipment to support the significant microbial community.
We carried out this study (summarized from the original publication in Aquacultural Engineering 77 (2017) 9–14) to compare CW and BF systems in terms of shrimp production, water quality dynamics and the estimated nutritional contribution of suspended biofloc particles in BF systems.
Study setup
This project was conducted at Kentucky State University’s Aquaculture Research Center (Frankfort, Ky., USA) in a building with sheet metal walls and a translucent, polycarbonate roof.
Six identical fiberglass tanks – each with an internal diameter of 153 cm, an operating depth of 74 cm, and a volume of 1.36 m3 – were arranged in two rows of three; three tanks were randomly assigned to a CW treatment and three to a BF treatment.
Pacific white shrimp (Litopenaeus vannamei) post-larvae (PL-12) were sourced from a commercial supplier (Shrimp Improvement Systems, LLC; Islamorada, Florida, USA). Before being stocked into the experimental tanks, the shrimp were raised in a clear-water nursery system for 30 days.

Please refer to the original publication or contact the first author for a detailed description of the experimental systems and experimental design, animal husbandry, water quality, use of stable isotopes to determine C and N incorporation into shrimp tissues, as well as the statistical analyses used in this study.

Results
Regarding water quality, water temperature, dissolved oxygen (DO) and salinity were all within acceptable ranges for Pacific white shrimp. Considering pH, it was relatively constant in the CW system but significantly lower in the BF system and possibly caused some stress in the animals.
The CW system had significantly lower turbidity, reflecting the additional mechanical filtration; it was higher in the BF system but it is not unusual to have high turbidity in biofloc systems.
Measured ammonia levels were regularly higher in the CW system, resulting in a significant difference between the treatments; however, all ammonia concentrations measured were below the estimated safe levels for L. vannamei.
Regarding shrimp production, the animals reached a significantly larger average size of 11.6 grams in the CW treatment compared to the 11.1 grams attained in the BF treatment (Table 1), paralleling the considerably greater biomass production in the CW systems. Feed conversion ratio in the CW system was 1.5:1, significantly better than the 1.8:1 in the BF system. Shrimp survival was not significantly different between the treatments, but mean survival in the CW system was markedly higher at 78 percent in contrast with 69 percent in the BF treatment, resulting in substantially better shrimp production in the CW system.
Ray, biofloc, Table 1
| Biofloc | Clear-water RAS | |
|---|---|---|
| Individual weight (g) | 11.1±0.2 (10.8-11.3)a | 11.6±0.3 (11.1-12.2)b |
| Biomass produced (kg/m3) | 1.7±0.0 (1.6-1.8)a | 2.0±0.1 (1.9-2.2)b |
| Growth rate (g/week-1) | 1.4±0.0 (1.4-1.4) | 1.5±0.0 (1.4-1.5) |
| FCR | 1.8±0.1 (1.7-1.8)a | 1.5±0.1 (1.3-1.6)b |
| Survival (%) | 69±0.6 (68-70) | 78±4.3 (70-85) |
The high particulate concentrations (as indicated by the turbidity values) in the BF treatments was a potential cause for their relatively diminished shrimp production. High particulate concentrations can increase the oxygen demand of the microbial community, obstruct the animals’ gills, encourage undesirable microorganisms and reduce shrimp growth.
Various studies in the scientific literature report that shrimp grown in water from ponds with particulate matter performed significantly better than shrimp grown in saline and clean well water, which contradicts our results. However, those studies were conducted at lower shrimp densities and particulate concentrations than the current study, and unlike ours, dense algal blooms and meio-fauna were present.
In addition, our results may be different from some previous studies because the nutritional contribution of natural biota like biofloc is reduced at higher animal density in more intensive systems, so that biofloc as a nutritional supplement may not be as important in intensive systems as it is in traditional semi-intensive ponds.
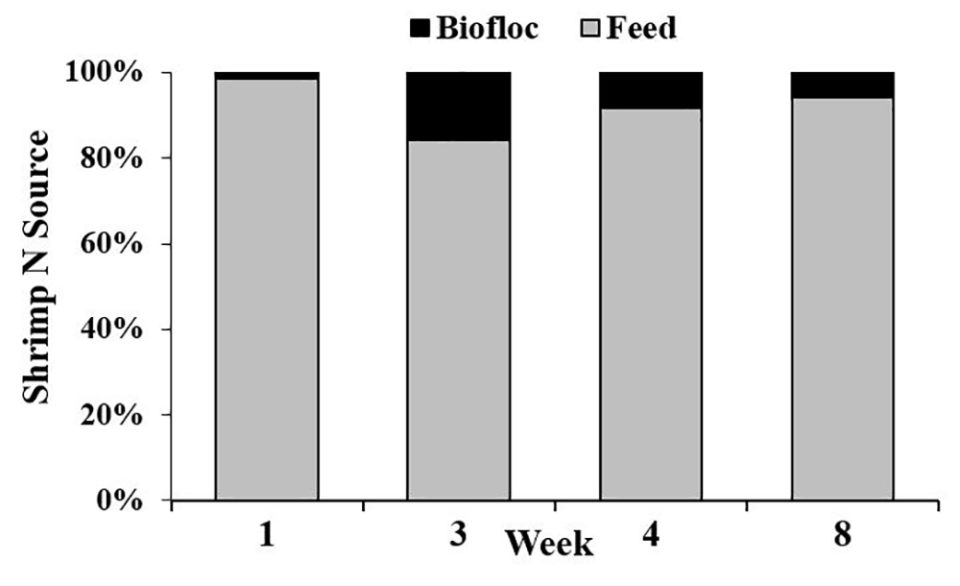
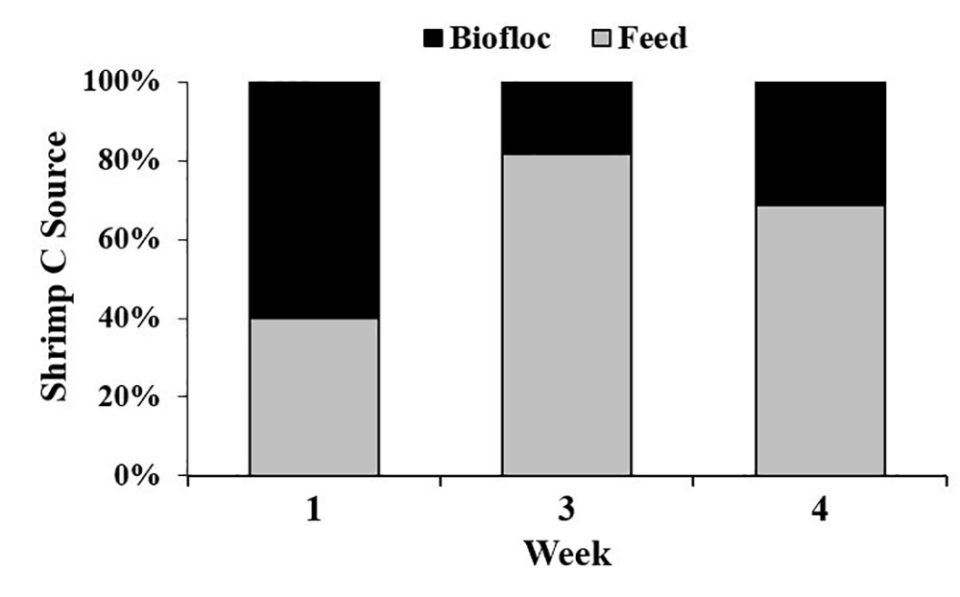
Regarding isotope dynamics, we examined the levels of C and N isotopes in shrimp, feed, and biofloc to obtain estimates of where shrimp are obtaining these elements. Isotopes are generally measured as a ratio of heavy/light, in this case C13/C12 and N15/N14, and these data are reported as a d-value in per mil units (‰); the exact formula is presented in the original publication.
Our results showed that there were no significant differences between shrimp d N15 values in the two treatments. However, animals from the CW treatment had significantly lower d C13 values vs. shrimp from the BF treatment, indicating that the animals in each treatment gained dietary carbon from different sources, and suggesting that biofloc may have been a source of carbon in the BF treatment.
Shrimp production in the BF system was not improved relative to shrimp in the CW system. In contrast to the findings of other related studies, biofloc may not have contributed much to shrimp growth in our study because the estimated contribution of N from biofloc to shrimp in this study was very small.
Perspectives

Results of our study show that CW tanks had significantly higher levels of ammonia and pH, and BF tanks had significantly higher nitrate, nitrite and turbidity levels. Based on stable isotope data, biofloc contributed 18 to 60 percent of the carbon and 1 to 16 percent of the nitrogen of the body tissue gained by shrimp. But these nutrient contributions from biofloc did not correspond to better shrimp production in the BF treatment, because shrimp total biomass, individual weights and FCR were all significantly better in the CW treatment vs. the BF treatment.
It is not clear exactly what led to these disparities in shrimp production, but the differences in water quality may have been involved. Our results indicate that, for indoor marine shrimp production, clear-water recirculating aquaculture systems may be a more productive option than biofloc systems.
References available from first author.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-

Andrew J. Ray, Ph.D.
Division of Aquaculture
Kentucky State University
Land Grant Program
103 Athletic Rd.
Frankfort, KY 40601 USA
[117,100,101,46,117,115,121,107,64,121,97,114,46,119,101,114,100,110,97]
-

Thomas H. Drury
Division of Aquaculture
Kentucky State University
Land Grant Program
103 Athletic Rd.
Frankfort, KY 40601 USA
-

Adam Cecil
Division of Aquaculture
Kentucky State University
Land Grant Program
103 Athletic Rd.
Frankfort, KY 40601 USA
Tagged With
Related Posts

Aquafeeds
Biofloc consumption by Pacific white shrimp postlarvae
The stable isotopes technique with δ13C and δ15N can be used to determine the relevance of different food sources to shrimp feeding during the pre-nursery phase of Litopenaeus vannamei culture. During this trial, different types of commercial feed, microalgae, Artemia sp. nauplii and bioflocs were used as food sources.

Intelligence
Biofloc technology production promising in temperate zones
A study was conducted to assess the feasibility to grow Channel catfish (Ictalurus punctatus) in an outdoor biofloc system during winter in a temperate zone. High biomasses of market-size channel catfish were successfully maintained through the winter with high survival and in good condition in both treatments.

Intelligence
An inside look at Sino Agro Foods’ giant prawn MegaFarm
Sino Agro Foods has developed a proprietary recirculating aquaculture system that yields high production volumes and profitability. The facility should significantly contribute to seafood production in China and to help satisfy increasing demand for high value, safe and sustainably produced seafood.

Health & Welfare
Aquamimicry: A revolutionary concept for shrimp farming
Aquamimicry simulates natural, estuarine production conditions by creating zooplankton blooms as supplemental nutrition to the cultured shrimp, and beneficial bacteria to maintain water quality. Better-quality shrimp can be produced at lower cost and in a more sustainable manner.


