Shrimp have difficulty molting if total alkalinity is less than 50 milligrams per liter

Inland shrimp culture in low-salinity water is an important activity in Thailand that accounts for about 30 percent of national production (Fast and Menasveta 2000). Shrimp are cultured in ponds containing water of 2 to 5 ppt salinity prepared by mixing brine solution from coastal seawater evaporation ponds with freshwater.
Because of the success with inland shrimp farming in Thailand, this practice is being attempted in Ecuador, Brazil, the United States, and other nations (Scarpa 1998, Samocha et al. 2001, Nunes and Lopez 2001), where saline groundwater from wells is often the source of low-salinity water (Boyd 2001). Some producers also mix granular salt from mines or evaporation ponds with freshwater to prepare low-salinity water.
Ionic composition and shrimp mortality

Major problems for inland shrimp farming outside Thailand have included high mortality of postlarvae (PL) during acclimation to low-salinity water and low survival in ponds during culture at some locations. In Ecuador, several farms with such problems sent water samples to Auburn University in Alabama, USA for analysis.
The water contained less than 10 milligrams per liter potassium. Fertilizer-grade potassium chloride (muriate of potash) was applied to increase potassium to 50 milligrams per liter, and shrimp survival improved. Analyses of water from ponds in Alabama with chronic shrimp mortality also revealed potassium concentrations of 5 to 15 milligrams per liter, and mortality declined after additions of muriate of potash.
Differences in ionic composition
Mortality associated with the ionic composition of water has not been reported in Thailand, where brine solution from seawater evaporation ponds is used almost exclusively for preparing low-salinity water for shrimp culture. Also, previous studies of the quality of groundwater in the area where inland shrimp farming is being attempted in Alabama revealed wide differences in ionic concentrations among wells located in close proximity (Boyd and Brown 1990).
It is reasonable to assume that shrimp require water with specific concentration ranges of major anions (bicarbonate and carbonate, sulfate, and chloride) and major cations (calcium, magnesium, potassium and sodium).
Test for ionic deficiencies
Laboratory trials could be conducted to determine acceptable concentrations of major ions for inland shrimp production (Laramore et al. 2001). However, until ionic requirements are established through research, a simple way of predicting the possible ionic deficiencies of a particular water for inland shrimp farming is to compare its ionic composition to that of waters that have proven suitable for shrimp culture.
Study objective
In a recent study supported by the Pond Dynamics/ Aquaculture Collaborative Research Support Program and funded by the U.S. Agency for International Development, the authors compared data on ionic concentrations in coastal and inland shrimp ponds, brine solutions, and granular salt from Thailand, Ecuador, and Alabama, USA. In Thailand, problems with ionic relationships have not been observed in widespread inland shrimp culture.
Sample collection
Water samples were collected from eight inland shrimp ponds and 17 wells in Ecuador during May 2001. In addition, 12 samples of brine solution and three samples of granular salt were collected from salt works.
Coastal shrimp ponds along the Gulf of Thailand between Nakon Sri Thammarat and Ranot, and inland shrimp farms in the vicinities of Rungsit, Chachoengsao, Bang Pakong, Ratchaburi, and Samut Songkhram were visited during mid-June 2001. Surface water samples were collected from 16 coastal shrimp ponds and 23 inland shrimp ponds. Samples of nine brine solutions and three granular salt samples also were obtained from supplies on farms or salt works.
Water samples were collected from eight wells and 11 ponds in Alabama, USA during May and November 2001.
Sample analysis
Samples were analyzed for major ions by the standard protocol recommended by Clesceri et al. (1998): calcium (titration to Eriochrome Black-T endpoint with ethylenediamine tetraacetic acid, EDTA), magnesium (titration to murexide endpoint with EDTA), sodium and potassium (atomic absorption spectrophotemetry), bicarbonate and carbonate (sulfuric acid titration), sulfate (barium chloride turbidimetry) and chloride (mercuric nitrate titration to diphenylcarbazone endpoint).
Salinity was determined with a salinity refractometer, and specific conductance was measured with a line-operated, research-grade conductivity meter. It is not possible to make accurate total dissolved solids (TDS) analyses of samples with a high salt content because salts retain water of hydration after evaporation (Stickland and Parsons 1972, Brown et al. 1989). About 95 to 99 percent of TDS in saline samples results from major ions. Thus, concentrations of major ions were summed to provide estimates of total dissolved solid concentrations (Boyd 2000).
Results
Ion concentration
The total concentration of major ions and concentrations of individual ions, except bicarbonate and carbonate, in coastal ponds were about one-half those of average seawater (Tables 1 and 2). Few samples contained measurable carbonate concentrations, for pH seldom exceeded 8.3. Thus, for convenience, the results of the bicarbonate and carbonate titration will be expressed in terms of bicarbonate in the rest of this article.
Total dissolved solids

intended for human and industrial uses.
Inland shrimp farming in Thailand, Ecuador, and Alabama is conducted in less-mineralized water than coastal shrimp farming in Thailand (Table 2). The average concentration of TDS was 18,539 milligrams per liter in coastal ponds, while averages for inland ponds ranged 3,888-8,739 milligrams per liter.
Bicarbonate concentrations were similar between coastal and inland ponds. Inland ponds in Ecuador averaged higher in calcium concentration than coastal ponds. Otherwise, average concentrations of individual ions were considerably higher in coastal ponds than inland ponds. Variation in concentrations of individual ions was great for all four data sets.
Waters of inland shrimp ponds in Thailand and Alabama were similar in TDS concentration, but the water in Alabama ponds had much lower average concentrations of magnesium, potassium, and sulfate. Inland pond water in Ecuador tended to have higher TDS concentrations and concentrations of calcium, magnesium, sodium, and chloride than water from ponds in Alabama and Thailand.
Aquifer water
Water from saline aquifers in Ecuador was more highly mineralized than that from saline aquifers in Alabama (Table 1), but well waters were less saline than average seawater and usually less saline than water from coastal ponds in Thailand. There was considerable variation among wells in the concentrations of TDS and individual ions, but Ecuadorian well waters tended to have greater concentrations of all ions than saline well waters in Alabama (Table 3).
Saline well waters in Alabama were especially deficient in potassium, magnesium, and sulfate as compared to those in Ecuador. Ponds filled with these well waters also had low concentrations of potassium, magnesium, and sulfate (Table 2).
Brine solutions
Brine solutions from Ecuador were more concentrated than those from Thailand (Table 1). Thailand samples were collected after the beginning of the rainy season, and dilution by rainwater had occurred. It is normal procedure to use brine solutions of 150 to 250 ppt salinity as a salt source for inland ponds in Thailand. Concentrations of individual ions also were slightly greater in Ecuadorian brine solutions than those from Thailand (Table 4).
Granular sal
Granular salt from Ecuador and Thailand had different concentrations of ions (Table 5). Ecuadorian samples were lower in potassium, magnesium, bicarbonate, and sulfate, but higher in other ions than the Thai samples. Ecuadorian salt was produced to maximize sodium chloride content and eliminate other ions, because it was intended for human consumption and industrial use. Thai salt samples were prepared for use in inland shrimp farming.
Measuring salinity
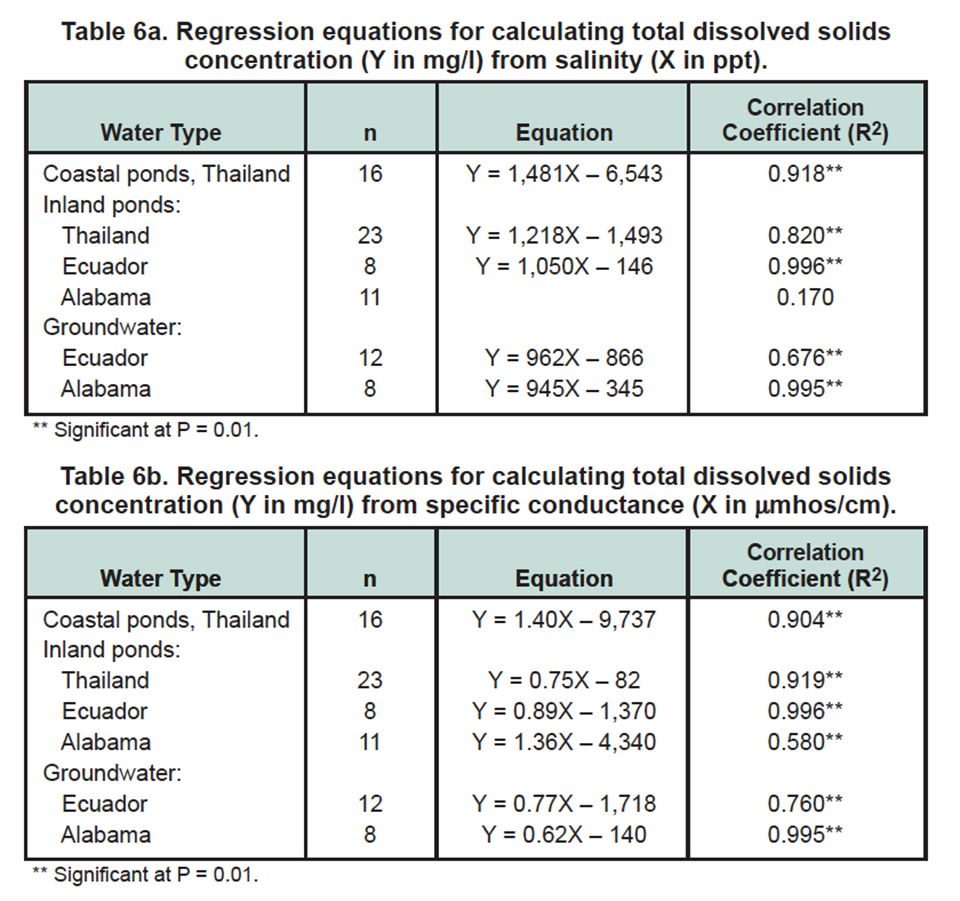
Salinity of shrimp pond water typically is measured with salinity refractometers. Since these devices are not highly accurate at low salinities of 0 to 5 ppt, Boyd (2002) suggested specific conductance as an alternative method for direct estimation of the degree of mineralization of water in inland shrimp ponds.
Both salinity and specific conductance were relatively good predictors of TDS concentration, but overall, specific conductance had a better correlation with TDS concentrations (Tables 6a, 6b). Data from inland shrimp ponds in Thailand suggests that shrimp can be cultured successfully in waters with specific conductance as low as 1,700 millimhos per centimeter, but average specific conductance was 5,704 μ millimhos per centimeter.
Degree of mineralization
Because of higher salt concentrations in coastal ponds, salinity refractometry is satisfactory for measuring the degree of mineralization. Salt concentrations in several brine solutions were too high to measure by conductivity meter or salinity refractometer. In Thailand, salt content of brine solutions is typically estimated from specific gravity as measured with a density hydrometer.
Acceptable ionic proportions
Seawater obviously has acceptable proportions of major ions for marine shrimp culture. Other sources of water, brine solutions, and granular salt can be evaluated based on the magnitude of deviation in ionic proportions from those of seawater.
To facilitate this comparison, the average contribution of each ion to TDS concentration (Sturm 1980) was calculated for each set of samples. The procedure was to divide the concentration of each major ion (grams per cubic meter) by TDS concentration (kilograms per cubic meter). To illustrate, seawater contains 34,462 grams per cubic meter or 34.462 kilograms per cubic meter of TDS. Calcium concentration in seawater is 400 grams per cubic meter, and the contribution of calcium to TDS is 400 grams Ca per cubic meter or 34.462 kg TDS per cubic meter = 11.6 grams Ca per kilogram TDS.
Ionic proportions (Tables 7a, 7b) reveal some interesting relationships. The contribution of individual ions to TDS is fairly constant between seawater and water in coastal shrimp ponds. This was expected for ponds filled from canals connected with the sea, and ponds within 2 km from the sea. Seawater was diluted with freshwater, but seawater provided most of the TDS and the proportions of each ion changed little. The slightly higher bicarbonate contribution to TDS in ponds was probably caused by the application of liming materials.
Calcium and bicarbonate
The contribution of calcium and bicarbonate to TDS was much greater in waters of inland shrimp farms than in seawater and waters of coastal shrimp farms (Table 7a). This difference exists because surface water and well water often have higher concentrations of bicarbonate than seawater, and because the contribution of calcium to TDS usually is much greater in freshwater than seawater (Hem 1970; Boyd 2000).
Liming materials also are applied to inland shrimp ponds to increase calcium and bicarbonate concentrations. The potassium contribution to TDS was less in inland shrimp ponds than in seawater and coastal ponds. However, the difference was considerably less for inland ponds in Thailand than those in Alabama and Ecuador. Sulfate contributed less to TDS in inland shrimp ponds in Ecuador and Alabama than in seawater, coastal ponds, and inland ponds in Thailand.
Contributions of individual ions
Brine solutions from Thailand and Ecuador were similar in ionic proportions except for a greater amount of sulfate in samples from Thailand and more bicarbonate in samples from Ecuador (Table 7).
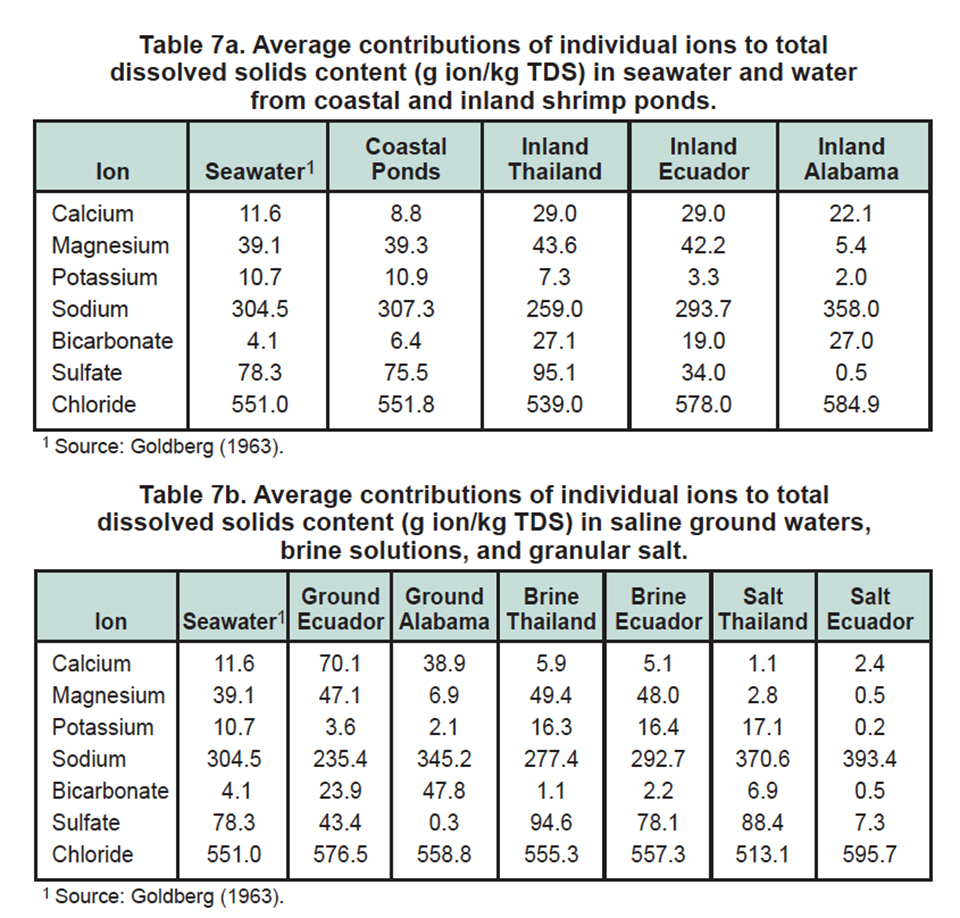
The amounts of individual salt ions relative to TDS were comparable to those of seawater except for less calcium and bicarbonate in brine. This difference is not considered important in shrimp ponds. Freshwater used to dilute brine will contribute calcium and bicarbonate, or ponds will be treated with liming materials that supply calcium and bicarbonate.
The TDS in groundwater from Ecuador and Alabama contained more calcium and bicarbonate than seawater (Table 7), but this groundwater had considerably less potassium and sulfate than seawater. The inland shrimp ponds in Alabama and Ecuador were supplied with groundwater, which explains why the TDS in pond water at these locations had lesser amounts of potassium and sulfate than seawater.
Granular salt samples from Ecuador consisted primarily of sodium and chloride (Table 7). A low-salinity water prepared by dissolving Ecuadorian salt in freshwater would not resemble seawater in relative proportions of ions. A low-salinity water made with salt from Thailand would have a shortage of calcium and magnesium as compared to seawater.
Suitable ionic proportions
Data in Table 7 suggests that mixing brine solution from seawater evaporation ponds with freshwater is the most reliable way of preparing low-salinity water with suitable ionic proportions for shrimp culture. Success achieved in Thailand with this practice confirms the conclusion.
During evaporation, brine solutions retain ionic proportions similar to those of seawater until salinity reaches about 250,000 milligrams per liter. Above this concentration, the differential precipitation of salts causes ionic proportions in the brine to change (Sturm 1980).
Brine solutions of around 200,000 milligrams per liter salinity are ideal for use in inland shrimp culture. Brines of this salinity have a high salt content to minimize volume requirements, while still assuring proportions of individual ions similar to those of seawater. Brines made by dissolving salt from salt deposits may not have the same composition as those from seawater evaporation ponds, and should not be used unless an analysis reveals they are suitable.
Granular salt may or may not be suitable for preparing low-salinity water for inland shrimp culture. For example, granular salt from Ecuador (Table 5) would be unsuitable, for it is mostly sodium chloride and would not supply adequate amounts of other ions. Granular salt from Thailand probably would be suitable, for it only has a shortage of calcium and magnesium.
These two ions will be present in the freshwater used to dilute the brine, and can also be supplemented by applying dolomitic limestone. In fact, some farms in Thailand use granular salt to maintain salinity in pond waters initially made saline with additions of brine solution.
Initial concentration may change
The salinity and ionic concentrations that initially result from mixing freshwater with brine solution, saline well water, or salt can be calculated. However, the initial concentration in the mixture may change in culture ponds for several reasons. Rainfall and runoff that enter ponds dilute ion concentrations and flush ions. This process lowers salinity, but should not alter ionic proportions because rainwater and runoff are dilute in their concentrations of ions (Boyd 2000).
Data from Ecuador and Alabama revealed that saline groundwater can have high concentrations of bicarbonate and calcium due to carbon dioxide supersaturation. When such water is held in a pond or other open system, carbon dioxide equilibrates with the air, and calcium carbonate precipitates. This causes a decline in the proportions of calcium and bicarbonate in the TDS.
Reactions with bottom soils

Reactions with pond bottom soil can also cause changes in the composition of low-salinity water in ponds. Negatively charged clay and soil organic matter can exchange cations with water and result in an equilibrium between the cations on exchange sites in bottom soils and pond water (Boyd 1995).
The influence of cation exchange between the bottom soil and water depends upon the cation exchange capacity of the bottom soil, concentrations of individual cations on exchange sites, soil pH, concentrations of individual cations in pond water, and rate of water mixing in ponds.
The influence can be considerable. Cation exchange with bottom soil can occur to a depth of 15 cm (Boyd and Cuenco 1980), and this layer can have a dry weight of about 150 kilograms per square meter (Munsiri et al. 1995). If bottom soil has a cation exchange capacity of 25 meq per 100 g, it could hold – and theoretically exchange – 37.5 eq cations per square meter. In a 1.5-meter-deep pond, this would be equal to 25 eq cations per cubic meter or several hundred milligrams per liter of cations (25 eq per cubic meter = 304 mg magnesium per liter, 501 mg calcium per liter, 575 mg sodium per liter, 978 mg potassium per liter).
Bottom soils can be acidic and neutralize bicarbonate in water. They can also contain deposits of limestone, gypsum, or other minerals that dissolve to increase the concentrations of ions in pond water.
Changes in ionic proportions
Examples of changes in ionic proportions in pond waters relative to brine solutions and saline groundwater used to prepare low-salinity water in ponds can be found in Table 7. The most striking change is the lower contributions of calcium and bicarbonate to TDS content in ponds in Ecuador and Alabama, as compared to well water used to fill ponds. The contribution of bicarbonate to TDS in pond water in Thailand is considerably greater than the contribution of these ions to TDS in brine solution.
Conclusion
The proportions of major ions in low-salinity water are no doubt important, but there is likely a requirement for minimum concentrations of some or all major ions. For example, shrimp have difficulty molting if total alkalinity is less than 50 milligrams per liter (61 milligrams per liter bicarbonate).
Shrimp can be produced in water with salinity of 1 ppt or possibly less (Jory 1999, Samocha et al. 2001), but acclimation is difficult. In Thailand, producers usually add enough brine solution to inland ponds to give an initial salinity of 5 to 6 ppt. Salinity gradually declines during the crop, often to only 1 to 2 ppt at harvest. Additional brine solution or granular salt can be applied if salinity falls below 1 to 2 ppt.
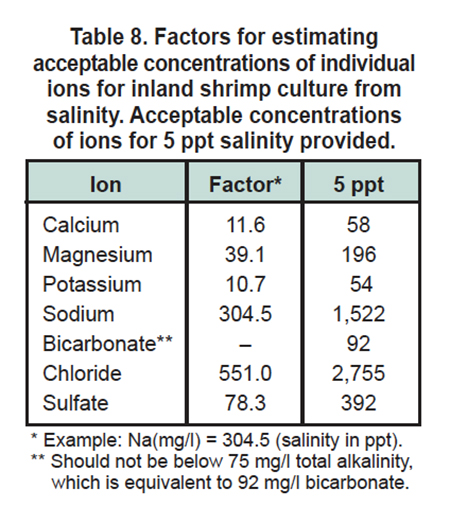 Until major ion requirements for shrimp are established in laboratory trials, it seems reasonable to consider 5 ppt as a minimum salinity requirement at the initiation of a shrimp crop. The minimum acceptable concentration of total alkalinity should be 75 milligrams per liter (about 90 milligrams per liter bicarbonate) (Boyd and Tucker 1998).
Until major ion requirements for shrimp are established in laboratory trials, it seems reasonable to consider 5 ppt as a minimum salinity requirement at the initiation of a shrimp crop. The minimum acceptable concentration of total alkalinity should be 75 milligrams per liter (about 90 milligrams per liter bicarbonate) (Boyd and Tucker 1998).
Minimum concentrations of other ions could be taken as concentrations expected in a solution prepared by diluting normal seawater to 5 ppt salinity (Table 8). The factors provided in Table 8 can be multiplied by ppt salinity to provide minimum concentrations of major ions at other salinity values.
Concentrations of individual ions can be enhanced in pond water by adding specific compounds. The ions of concern are bicarbonate, calcium, magnesium, potassium, and sulfate. Sources of these ions are as follows: bicarbonate (total alkalinity); agricultural limestone (CaCO3 or MgCO3 × CaCO3); potassium, muriate of potash (KCl) or potassium sulfate (K2SO4); magnesium, dolo-mitic limestone (MgCO3×CaCO3) and magnesium sulfate (MgSO4); sulfate, sulfuric acid (H2SO4), potassium sulfate or magnesium sulfate. These compounds are widely available and relatively inexpensive.
Sometimes, low-salinity water from wells can be used to acclimate postlarvae. This water must have adequate concentrations of ions, and precipitation of mineral matter should not occur. Calcium carbonate precipitation that can occur from high-alkalinity well water has been observed to form harmful encrustations on PL in Thailand (Chalor Limsuwan, personal communication).
Some well water in Ecuador contained 20 to 30 milligrams per liter iron and 10 to 20 milligrams per liter manganese. When such water is aerated, the precipitation of iron and manganese oxides and hydroxides can harm PL. Water that is expected to yield precipitates should be held several days to encourage oxygenation, loss of excess carbon dioxide, and sedimentation of precipitates before shrimp are stocked. Mechanical aeration can accelerate the oxidation of iron and manganese, and carbon dioxide removal. In acclimation facilities, aeration and filtration can be used to accelerate the removal of precipitates (Tucker 1988).
Note: Cited references are available from the first author.
(Editor’s Note: This article was originally published in the June 2002 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Authors
-

Claude E. Boyd, Ph.D.
Department of Fisheries and Allied Aquacultures
Auburn University
Auburn, Alabama 36849 USA[117,100,101,46,110,114,117,98,117,97,64,49,101,99,100,121,111,98]
-
Taworn Thunjai
Department of Fisheries and Allied Aquacultures
Auburn University
Auburn, Alabama 36849 USA -
Mali Boonyaratpalin, Ph.D.
Department of Fisheries
Ministry of Agriculture and Cooperatives
Bangkok, Thailand
Related Posts

Responsibility
A review of water quality improvement products
Prof. Boyd examines products used by aquafarmers to improve water quality and conditions in their ponds and discusses their efficacy.

Aquafeeds
Alternatives to fishmeal perform well in low-salinity shrimp farm trial
In a white shrimp farm trial in a low-salinity environment, alternatives to fishmeal did not negatively impact the growth, survival or FCR of the shrimp.

Responsibility
Assessing groundwater quality in aquaculture
Those interested in using groundwater for aquaculture should perform a thorough chemical analysis of the water. Several problems related to groundwater use in hatcheries and holding or transport vessels can be alleviated by degassing or aeration.

Responsibility
Calcium and magnesium use in aquaculture
Aquatic plants and animals get the essential nutrients calcium and magnesium from water and food. Calcium concentrations impact the hydration and development of eggs in a hatchery, where calcium carbonate precipitation can be troublesome.



