Results show potential for salinity-specific breeding programs for ammonia tolerance

Due to the advantages of its euryhalinity (tolerance to broad salinity ranges), rapid growth rates and a high survival rate at high stocking densities, the Pacific white shrimp (Litopenaeus vannamei) is the world’s most popular cultured shrimp species.
High concentrations of ammonia are the most common and greatest toxic factor in shrimp culture ponds. Increased ammonia in the water can suppress the immune defense system of shrimp and increase their sensitivity to pathogens. We previously carried out a study to understand the molecular mechanisms of the detrimental effects of ammonia stress in L.vannamei and determined that the majority of the key genes involved in the response to ammonia stress are potentially involved in immune defense function.
Previous studies have revealed that ammonia stress is more toxic to the earlier ontogenetic development of aquaculture organisms, and it has been widely found to exert lethal effects on penaeid shrimp juveniles. Early juvenile shrimp are critical for successful breeding because high rates of mortality often occur during this period. And the phenotype (an organism’s observable characteristics or traits) of a quantitative trait (phenotypes that are controlled by multiple genes) is determined by genetic and environmental resources and their interactions; thus, genotype-by-environment (G×E) interactions will play an important role in genetic improvement.
L. vannamei is cultured at different salinity levels due to its euryhalinity, but several studies have revealed that low salinity can increase the toxicity of ammonia to the shrimp, and we have also verified that ammonia stress can influence the pathway that is involved in osmoregulation. If the G×E interaction for ammonia tolerance is significant at different salinity levels, the selection response for this trait will vary across different salinity levels. Based on this, it is necessary to understand the heritability of ammonia tolerance in the early stage and the G×E interactions at different salinity levels in L. vannamei.
This article – adapted and summarized from the original publication – reports on a study to estimate the heritability of ammonia tolerance in L. vannamei juveniles in 30 ppt and 5 ppt salinity. We estimated the heritability of ammonia tolerance in L. vannamei juveniles in the early stage (average body weight, 0.5 grams) at two different salinity levels (30 and 5 ppt) and estimated the genetic correlations between body length and ammonia tolerance within the two environments to verify the relationship between ammonia tolerance and ontogenetic development.
This work was supported by the Central Public-interest Scientific Institution Basal Research Fund, CAFS (No. 2016HY-ZD04), the National Natural Science Foundation of China (No. 31302179), the Taishan scholar program for seed industry, the Introduction of International Advanced Agricultural Science and Technology Plan (No. 2016-X39), and the China Postdoctoral Science Foundation (No. 2015M572095).
Study setup
This study was carried out at a Hebei Xinhai Aquatic Biological Technology Co., Ltd. facility in Huanghua, Hebei Province, China. The founder animals were collected from seven improved commercial strains that were introduced from different companies in the United States and Singapore. The virus-free individuals from the seven strains were used to produce the base population (G0) with 130 full-sib families using artificial insemination with 114 males and 108 females and included 69 half-sib families. Families with low selection indices were eliminated from the breeding program, and the males and females with high selection indices from the remaining families were selected to produce the next generation.
Experimental shrimp were from 91 full-sib families (52 half-sib families) of the fifth generation (G5). At approximately 45 days, around 400 postlarvae were randomly selected from each family and placed into tow-net cages (0.5 cubic meters) that were separately fixed in two large ponds (60 cubic meters) for separate rearing at the same density for each family. The seawater in one of the ponds was diluted with filtered fresh water from 30 ppt to 5 ppt, which lasted for approximately three weeks. However, the seawater in the other pond was maintained at 30 ppt. Aside from the salinity, other management conditions were maintained identically between the two ponds.
For detailed information on the origin of the base population and selection procedure; the challenge with high concentration ammonia; and the statistical analyses, including the variance components and heritability estimates; and the genotype by environment interaction estimate, consult the original publication.
Results and discussion
In this study, we first report the heritability of ammonia tolerance (adversity-stress adaptability) for juveniles in the early stage in normal salinity (30 ppt) and low salinity (5 ppt) conditions and the G×E interaction between the two environments to explore the possibility of reducing the mortality of shrimp by selection. Many studies have reported that ammonia stress is more toxic to shrimp in early stages [28–30, 53–55], and this finding was corroborated in the present study.
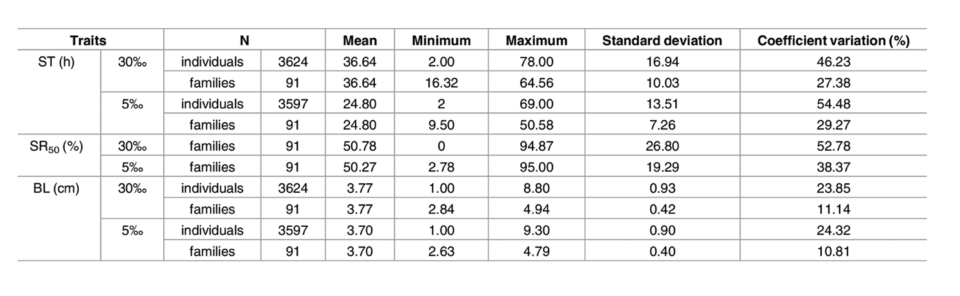
A total of 7,221 individual records, including 3,624 records from the normal salinity condition and 3,597 records from low salinity condition, were obtained and analyzed in the present study. The number of final testing individuals was less than the total number of starting individuals (7,280), which might have been due to cannibalism during the experimental process. The numbers of samples, simple means, minima, maxima, standard deviations and the coefficients of variation for the survival rate of each family at the half-lethal time (SS50), survival times (STs), and body length (BL) are given in (Table 1). The 25th percentiles, median percentiles, 75th percentiles, minima, maxima, and outliers of the SS50 and average ST values of each family are displayed in Fig. 1.
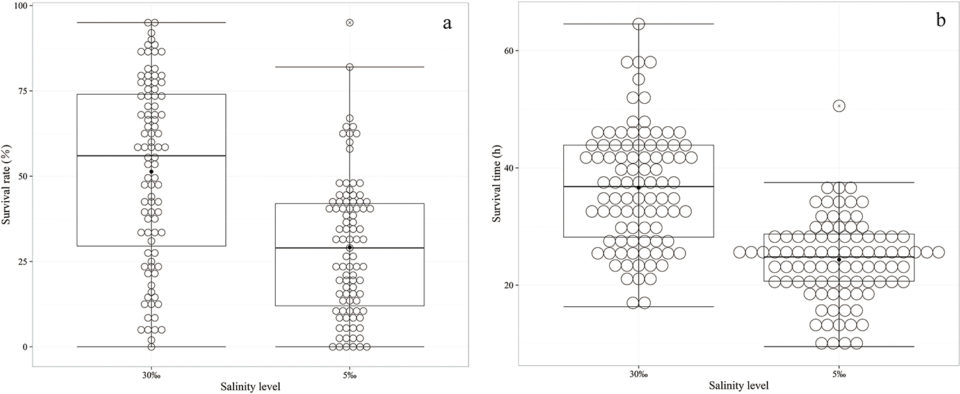
The results revealed the SS50 values under acute ammonia stress in the normal and low salinity conditions varied substantially between the families, but the variance was higher following exposure to acute ammonia stress in the normal salinity condition. Additionally, the survival times under acute ammonia stress in the normal and low salinity conditions also varied substantially between the families and the overall individuals, but the variance was higher when analyzed at the individual level than the family level as indicated by the higher standard deviation (SD) and coefficient of variation (CV).
The CVs of the STs were 27.38 percent and 29.27 percent among the 91 families in the two environments, but these values were 46.87 percent and 54.48 percent among the individuals in the two environments. Although the concentration of ammonia (32 mg/L) was higher in the normal salinity condition than in the low salinity (18 mg/L) condition, the average ST under acute ammonia stress in the normal salinity condition (36.64 hours) was substantially greater than that in the low salinity condition (24.80 hours), and the variance was higher in the normal salinity condition. Regarding the BL, it also exhibited higher variance when analyzed at the individual level than at the family level, but there was no significant difference between the two environments.
The cumulative mortality and the average survival time of each family under acute ammonia stress in the normal and low salinity conditions are displayed in Fig. 2 and Fig. 3, respectively. The cumulative mortalities of the families under acute ammonia stress in the low salinity condition were substantially greater than those in the normal salinity condition at each sampling point (Fig. 2). All of the individuals in the low salinity condition were dead after 69 hours of exposure to acute ammonia stress, but at this time, 115 individuals (3.5 percent) in the normal salinity condition were still alive. It showed that the average survival time exhibited large variation between the families within the same environment and between the two environments, but the average survival time of the majority of the families under acute ammonia stress in the low salinity condition was substantially lower than that of the families in the normal salinity condition (Fig. 3).
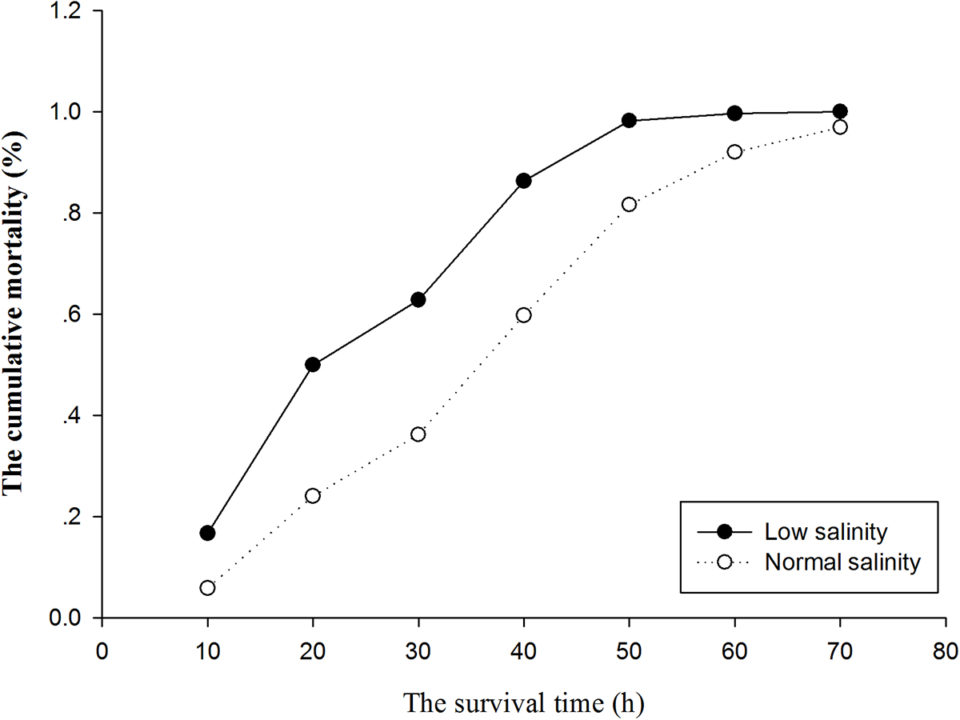
Although high temperatures and low salinities can increase the toxicity of ammonia stress, the shorter survival time at lower concentrations of ammonia in our study also suggests that younger shrimp are more sensitive to ammonia toxicity. Additionally, we also observed that the lower salinity increased the sensitivity of shrimp to the toxic effects of ammonia. The lethal concentration of ammonia at 72 hours in the normal salinity condition (30 ppt) was 32 mg/L, which is significantly higher than the 18 mg/L value observed in the low salinity condition (5 ppt).
The average survival time of all the individuals under acute ammonia stress in the normal salinity condition was 36.61 hours, which is substantially greater than the 24.80 hours observed in the low salinity condition, and the average survival time of majority of the families under acute ammonia stress in the normal salinity condition was significantly greater than that in the low salinity condition. The cumulative mortalities of the families in the normal salinity condition were significantly lower than those in the low salinity condition at each sampling point. These results indicated that low salinity increased the toxicity of ammonia stress to shrimp, which is consistent with the results of previous studies. Our results suggest that the selection of ammonia-tolerant shrimp should be performed in the early stage and in low salinity water.
We estimated the heritability of ammonia tolerance of shrimp with an average body weight of 0.5 grams under ammonia stress in normal and low salinity conditions. The heritability estimates under ammonia stress at the two salinity levels were all significantly greater than that reported in the previous study, which indicates that improving the ammonia tolerance of shrimp via selection performed at the early stage could be quite advantageous.
After this experiment, a white spot syndrome virus (WSSV) infection experiment was performed with the ammonia-sensitive and ammonia-tolerant populations to detect the relationship between ammonia tolerance and WSSV resistance. The results revealed that the ammonia-tolerant population also exhibited a significantly greater resistance to WSSV than the ammonia-sensitive population. The ammonia-tolerant shrimp can be improved largely due to high heritability, which might provide an alternative method to reduce mortality, aid the enhancement of resistance to pathogens and reduce infectious diseases.
We detected a strong positive correlation between body length and ammonia tolerance consistent with previous studies. The strong positive correlation between body size and ammonia tolerance suggests that it is necessary to select ammonia-tolerant shrimp during the early stage and that selection high growth will not adversely affect ammonia tolerance performance in shrimp.
G×E interactions refer to genotypes that respond differently in different environments and thus result in different performances. Genetic correlations across environments that are significantly different from 1 indicate the existence of re-ranking effects, i.e., genotypes that should be ranked differently in different environments. Generally, selection for a trait in aquaculture is always performed in the main environment; for example, ammonia tolerance would typically be selected for in normal salinity conditions. However, the offspring of the excellent shrimp procured from selection are transferred to different commercial production environments in most cases.
In our study, we detected a strong re-ranking effect (i.e., a low genetic correlation) for ammonia tolerance between the different salinity levels, which suggests that the genetic advantage of ammonia tolerance can be significantly influenced by salinity. Additionally, the genetic correlations across environments would decrease along with increases in differences between environments. When a strong G×E interaction exists for a trait, it is better to perform selection in multiple environments, such as the nucleus environment and other production environments. Therefore, salinity-specific breeding programs for ammonia tolerance in shrimp should be purposefully implemented.
Perspectives
Based on our results, we first report the heritability of ammonia tolerance in juveniles in the early stage and the G×E interaction between the normal and low salinity, to explore the potential for improving ammonia tolerance in shrimp by selection. The results revealed that the heritability of ammonia tolerance was medium to high in L. vannamei juveniles, which suggests that rapid genetic gains in terms of ammonia tolerance could be obtained by increasing the selection intensity in our selective breeding program. However, the heritability of ammonia tolerance in the normal salinity condition was significantly higher than that in the low salinity condition.
Additionally, a strong G×E interaction for ammonia tolerance was detected between the normal salinity and low salinity conditions, which suggests that salinity-specific breeding programs for ammonia tolerance in shrimp should be purposefully implemented and that the normal salinity environment is a better choice based on the faster rate of genetic improvement due to higher heritability.
Also, the significantly and strong positive correction between ammonia tolerance and body size suggests that ammonia-tolerant shrimp should be selected in the early stage.
References available from corresponding author.
Editor’s note: This article has 11 co-authors (mentioned in the keywords section below), but only the affiliation and contact information for the corresponding author is listed.
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Dr. Jie Kong
Key Laboratory of Sustainable Utilization of Marine Fisheries Resources
Ministry of Agriculture
Yellow Sea Fisheries Research Institute
Chinese Academy of Fishery Sciences
Qingdao, China
Laboratory for Marine Fisheries Science and Food Production Processes
Qingdao National Laboratory for Marine Science and Technology
Qingdao, China[110,99,46,99,97,46,105,114,102,115,121,64,101,105,106,103,110,111,107]
Tagged With
Related Posts

Health & Welfare
Ammonia addition enhances microbial flocs in nursery phase for Pacific white shrimp
In a study, “pre-fertilization” in the nursery phase of a biofloc system for shrimp was tested. The objective was to accelerate the biofloc formation to minimize ammonia concentrations, avoiding high peaks during culture.

Health & Welfare
Ammonia toxicity degrades animal health, growth
Ammonia nitrogen occurs in aquaculture systems as a waste product of protein metabolism by aquatic animals and degradation of organic matter, or in nitrogen fertilizers. Exposure can reduce growth and increase susceptibility to diseases in aquatic species.

Responsibility
Ammonia nitrogen dynamics in aquaculture
The major sources of ammonia in aquaculture ponds are fertilizers and feeds, and problems with high ammonia are most common in feed-based aquaculture.

Health & Welfare
Biofloc trial results in fast shrimp growth, low FCR, high survival
A trial in a lined, greenhouse-enclosed raceway evaluated the use of a heterotrophic biofloc system equipped with aeration, supplemental oxygen injection and centralized heating to achieve good shrimp production.


