Successful diet developed for freshwater prawns
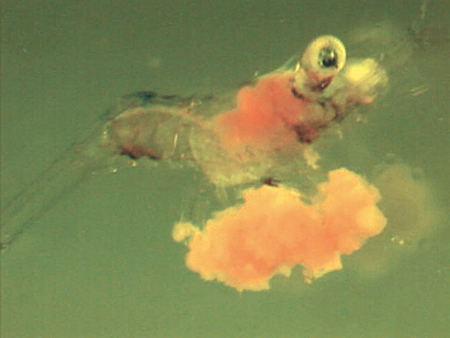
During the past five years, the Mississippi Alabama Sea Grant Consortium funded a research project by the author to develop a larval diet that would replace artemia as food for larval fish and crustaceans. After numerous modifications during experiments with the Malaysian freshwater prawn (Macrobrachium rosenbergii), a successful diet has been developed.
Artemia replacement
The ultimate goal of the project was to produce a formulated diet that would serve as a complete replacement for live food by achieving similar production characteristics at lower cost.
Cyst availability
The culture of larvae of many species of fish and crustaceans is precariously dependent upon the availability of artemia cysts from which the food, artemia nauplii, is obtained through hatching. The availability of these cysts from the natural environment is subject to periodic, unpredictable shortages. As a result, demand cannot always be supplied, and prices increase. The hatching of cysts is labor-intensive and also adds operational costs.
Nutritional quality
In addition, variations in the nutritional quality of hatched artemia nauplii relative to season and locale of harvest have been a chronic problem. This has been partly overcome by methods to enhance the nutritional value of the artemia, particularly as it relates to the content of two highly unsaturated fatty acids, docosahexaenoic acid and eicosahexaenoic acid.
Previous diets
Past attempts to produce nutritionally complete larval diets are commonly characterized as microencapsulated, microbound, or microcoated diets. To date, they have had limited success and, in most cases, not served as complete artemia substitutes.
Most of the success has been with herbivorous forms of larvae, where the rapid transit time through their guts did not require a highly digestible diet. Lack of success with carnivorous larvae was attributed to a long gut retention time and the possible absence of sufficient digestive enzymes during early development due to the lack of a stomach.
Initial attempts in this new research met the same frustrations encountered by other investigators over the years. The prepared diets were readily consumed and the animals’ guts were filled, but growth and survival rates were far below those observed for larvae fed live artemia nauplii. These observations suggested that either or both digestion and assimilation were poor.
Enzymes
The need for exogenous digestive enzymes in the diets was considered, but recent research strongly suggested a sufficient complement of enzymes was available during early development of fish and crustacean larvae. The inefficient digestion was probably not due to a lack of enzymes, but the quality of the enzymes relative to their capacity to effect digestion of specific protein sources.
Microbound diet
The successful microbound diet substitutes completely for live artemia nauplii during the larval metamorphosis of M. rosenbergii from stage 5 through 11 and then to postlarva. Although not experimentally documented, part of its success may be attributed to protein quality. Some protein sources have an attractive essential amino acid profile but are not efficiently digested.
Principally bound with alginate, the diet was designed to have high moisture content of about 60 percent. Presently, the high level of moisture precludes the production of particles less than 250 μ in size. However, in a dry form, the particle size can be reduced to 125 μ.
Larvae growth and survival
This is an energy-dense diet with 46 percent protein and 37 percent lipid levels. Survival of larvae fed the microbound diet exclusively for 14 days (23 days post-hatch) was 75 percent. Growth and survival of the larvae fed the microbound diet were 85 to 90 percent of that achieved with the artemia-fed larvae.
Easily modified
The most attractive characteristic of the diet is that all the ingredients are chemically defined. It does not contain any exogenous enzymes. Therefore, modification to address specific nutritional requirements of other species can be readily accomplished.
High acceptability
Another important quality of the diet is its high acceptability. In the past, experimental protocol in the testing of formulated larval diets has generally included a period of weaning from live diets. The new microbound diet does not require a weaning period. Upon first presentation in the absence of live food, this diet was immediately ingested by the M. rosenbergii larvae.
Water stability
The diet is water-stable, losing only about 5 percent of its dry weight after 1.5 hours of immersion with aeration. Most of this loss appears to be due to leaching of water-soluble nutrients. For example, 30 to 40 percent of the thiamin and riboflavin provided in a vitamin mix is lost during the first 15 minutes of immersion.
Nutrient overcompensation
To prevent the possibility of not satisfying nutritional requirements for water-soluble nutrients due to leaching, these nutrients are included in the diet at levels much higher than the reported requirements for juveniles.
This approach is particularly effective for species like Macrobrachium, which grasp particles with their feeding appendages as they ingest the diet. Some of the loss due to leaching would be reduced in the culture of carnivorous fish due to their more rapid ingestion of food as it enters the water column.
Conclusion
Although successful with M. rosenbergii, the potential of the microbound diet as an artemia substitute for other species is currently under investigation. Development and evaluation of the diet have so far been limited to small-scale conditions. The results need to be verified in commercial-size production units.
The diet should offer a final cost competitive with artemia cysts, as well as the added advantage of complete control and reproducibility of the nutrient composition of the diet through its patent-pending production process.
(Editor’s Note: This article was originally published in the August 2002 print edition of the Global Aquaculture Advocate.)
Now that you've reached the end of the article ...
… please consider supporting GSA’s mission to advance responsible seafood practices through education, advocacy and third-party assurances. The Advocate aims to document the evolution of responsible seafood practices and share the expansive knowledge of our vast network of contributors.
By becoming a Global Seafood Alliance member, you’re ensuring that all of the pre-competitive work we do through member benefits, resources and events can continue. Individual membership costs just $50 a year.
Not a GSA member? Join us.
Author
-
Louis R. D’Abramo, Ph.D.
Department of Wildlife and Fisheries
Mississippi State University
Mississippi 39762 USA[117,100,101,46,101,116,97,116,115,115,109,46,114,102,99,64,111,109,97,114,98,97,100,108]
Tagged With
Related Posts

Health & Welfare
A holistic management approach to EMS
Early Mortality Syndrome has devastated farmed shrimp in Asia and Latin America. With better understanding of the pathogen and the development and improvement of novel strategies, shrimp farmers are now able to better manage the disease.

Health & Welfare
Advances in intensive copepod production technology
Research at the Oceanic Institute has been successful in overcoming bottlenecks associated with rearing small-mouthed fish larvae by finding a suitable first feed. Early work on the calanoid copepod Parvocalanus crassirostris focused on parameters necessary for successful maintenance of stock cultures.

Innovation & Investment
Aquaculture America 2017: Communication key to the future
This year’s Aquaculture America in San Antonio, Texas, provided significant learning and networking opportunities. It successfully brought together 14 U.S. aquaculture organizations and more than 1,600 participants from Europe, Asia, Africa and Australia.

Health & Welfare
Artemia bioencapsulation delivers probiotics via digestive tracts of fish larvae
Research with sturgeon and carp species indicated that encapsulated artemia has high potential to carry probiotics or other beneficial microorganisms.


