In situ hybridization and PCR effective at detecting microsporidian parasite

In penaeid shrimp, microsporidia infect host tissues such as muscle, heart, gonads, gills, hepatopancreas, and nerve ganglia, depending on the type of microsporidia. Shrimp microsporidia are considered a potential threat for their populations, and also lead an economic impact in aquaculture. Recently, a microsporidium, Enterocytozoon hepatopenaei (EHP), that replicates within the cytoplasmic area of the tubule epithelial cells in the hepatopancreas has been found in several shrimp farming countries in Asia including Vietnam, Thailand, Malaysia, Indonesia, China and India. The clinical signs of the EHP infection are not specific, and associated with growth retardation.
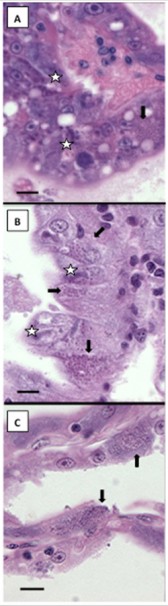
Histopathology of EHP
Histological examination of samples of EHP-infected Penaeus vannamei from Vietnam (2014) showed basophilic inclusions within the cytoplasm of hepatopancreas tubule epithelial cells of (Fig 1A & 1B). These inclusions appeared to be at plasmodia stage; mature, basophilic, spores were also observed. The EHP-infected P. stylirostris collected in Brunei (2006) were also showed the presence of microsporidium spores within the hepatopancreas (Fig. 1C). These results also indicate that EHP has been present in Asia since 2006.
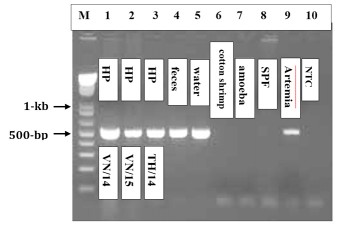
EHP PCR assay
We selected primers for detecting the EHP are EHP-510F (5’-GCCTGAGAGATGGCTCCCACG) and EHP-510R (5’-GCGTACTATCCCCAGAGCCCG) and PCR was performed with DNA extracted from EHP-infected shrimp. The results showed that this PCR can detect EHP-infected shrimp (Fig. 2, lane 1-3); the feces and water samples collected from infected shrimp tanks were also detected with EHP (Fig. 2, lanes 4 & 5). We also tested 2 other parasitic pathogens, an amoeba and the cotton shrimp disease microsporidium, but these EHP-primers did not cross react with them (Fig. 2, lane 6 & 7).
Unexpectedly, we detected EHP in 4 out of 9 samples of frozen Artemia biomass (Fig. 2, lane 9, a representative sample). From EHP-positive Artemia biomass, its 18S rRNA gene was amplified and sequenced, and the fragment was 99.9 percent identical to that of EHP from Vietnam, suggesting the EHP present in Artemia biomass may have originated from Southeast Asia.
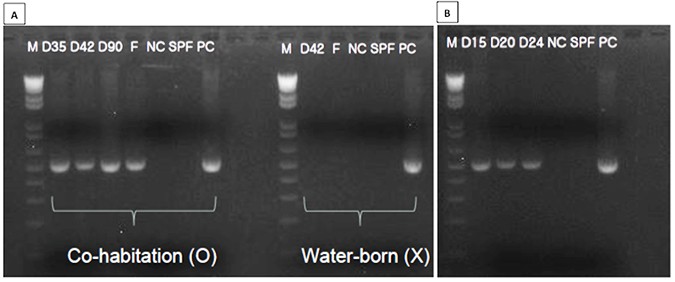
Studies of EHP transmission
Three bioassays were conducted using a population of EHP-infected shrimp sent from Thailand. In bioassay no. 1 (co-habitation group), diseased shrimp were transferred into a tank stocked with SPF shrimp tagged with visible elastomers. The SPF shrimp became infected after 35 days of co-habitation (Fig. 3A). For bioassay no. 2 (water-born infection group), only water was transferred from diseased shrimp tank into the SPF shrimp tank. The SPF shrimp were not affected even after 42 days (Fig. 3A). For bioassay no. 3, SPF shrimp were fed the hepatopancreas from the diseased shrimp. Samples were analyzed by PCR analysis every 5 days; SPF shrimp became infected at day 15 (Fig. 3B), suggesting that EHP can be transmitted via cannibalism.
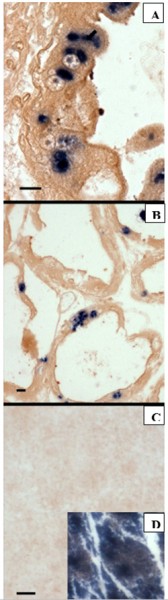
In situ hybridization (ISH)
By ISH with a digoxigenin-labeled EHP probe, the infected P. vannamei (Fig 4A) and the P. stylirostris (Fig 4B) showed the presence of microsporidium spores (basophilic inclusions) within the hepatopancreas, the tubule epithelial cells. The probe appears to be highly specific. No reaction was seen with Perezia penaei-infected P. monodon showing a clinical sign of cotton shrimp disease (Fig 4C).
Perspectives
Concerns of EHP infections in farmed shrimp populations are likely to continue, creating a need to reduce risk through the establishment of effective means to control and monitor this parasite. In this regard, the use of specific and sensitive molecular methods for the detection of EHP in shrimp, live feeds, and the pond environments will likely prove to be very important. EHP-infected shrimp cannot be determined by simple visual inspection; there are no obvious clinical signs of infection. Diagnostic protocols, based on PCR and ISH, developed in this study have proven to be both specific and sensitive, thus providing valuable tools for routine diagnosis and monitoring of shrimp stocks, pond environments and aquaculture commodities.
Authors
-

Jee Eun Han, Ph.D.
Postdoctoral Research Associate
School of Animal and Comparative Biomedical Sciences
University of Arizona
Tucson, AZ 85721 USA[117,100,101,46,97,110,111,122,105,114,97,46,117,64,110,97,104,101,101,106]
-

Kathy F.J. Tang, Ph.D.
Associate Research Professor
School of Animal and Comparative Biomedical Sciences
University of Arizona
Tucson, AZ 85721 USA[117,100,101,46,97,110,111,122,105,114,97,46,108,105,97,109,101,64,117,121,106,103,110,101,102]
-

Donald V. Lightner, Ph.D.
Professor Emeritus
School of Animal and Comparative Biomedical Sciences,
University of Arizona,
Tucson, AZ 85721 USA[117,100,101,46,97,110,111,122,105,114,97,46,117,64,108,118,100]
Related Posts

Health & Welfare
Limited decomposition enhances PCR detection of AHPND Vibrio in shrimp
A study confirmed the utility for improved polymerase chain reaction (PCR) detection of the Vibrio bacteria that cause acute hepatopancreatic necrosis disease (AHPND) in asymptomatic shrimp by permitting the shrimp to expire and decompose for several hours prior to preservation and PCR processing.
Health & Welfare
Double-stranded RNA against WSSV genes provides antiviral protection in shrimp
Silencing genes in white spot syndrome virus (WSSV) with critical roles in replication could provide a strong antiviral effect and thus reduce shrimp mortality. The authors therefore established a study to evaluate the antiviral efficacy of double-stranded (ds)RNA against non-structural WSSV genes.

Health & Welfare
A holistic management approach to EMS
Early Mortality Syndrome has devastated farmed shrimp in Asia and Latin America. With better understanding of the pathogen and the development and improvement of novel strategies, shrimp farmers are now able to better manage the disease.

Responsibility
CP aquaculture expert stresses proper pond management, design
Shrimp farmers can look to the chicken-farming industry for guidance on proper pond management. At the Nicovita Symposium in Ecuador, Dr. Dean Akiyama of Charoen Pokphand encouraged shrimp producers to overcome challenges at hatchery level.

