It’s important to monitor the proportioning, grinding, mixing, pelleting and packaging for in-line quality assurance

The starting point for quality feeds is quality ingredients. However, starting with high-quality ingredients does not ensure high-quality feeds unless strict guidelines are implemented at each of the processing cost centers. Therefore, assuring that the ingredients are processed properly into a high-quality product requires that each step of the process is carefully monitored to produce consistent quality shrimp aquafeeds.
In each of the cost centers (proportioning, grinding, mixing, pelleting, packaging) at an aquafeed plant there is a process and a product generated. The product can be defined as a blend of ingredients, the ground ingredients, the pelleted feed, the crumbles, and finally the bagged feed.
Optimization of each process
The first step for in-line quality monitoring is the optimization of each of the processes used to make the aquafeed. The objective of this optimization is to define the processing parameters where a maximum throughput is achieved to produce the highest possible product quality. The latter is a concept sometimes difficult to grasp by some plant and production managers who, in most cases, pursue increasing capacity – and not quality – as the measure of plant productivity.
In reality, the measure of productivity of any feed mill should be based on the highest quality at the maximum capacity, quality being the driving force, and not the other way around.
Process optimization can provide the guidelines or range of processing parameters that the equipment operator can use to consistently produce, every day, the highest quality feed possible. It can also be used as a tool to determine the robustness of the process, or how sensitive is the process to the range of processing conditions.
In other words, a small change in the processing parameters can produce the expected or measurable effect on the product quality. Therefore, the processing parameters or process optimization are the starting point for ensuring high quality aquafeeds.
Personnel commitment
For a successful in-line quality assurance program it is also necessary to have the commitment of all plant personnel. This can be achieved by providing all personnel and operators with the proper training about their equipment performance and how it can affect quality. The operators should also have full ownership and responsibility of the equipment to make sure that it is being maintained and operated properly.
No quality assurance program will work if the company does not endorse it through strict guidelines that should be strictly enforced by the Quality Assurance (QA) department, which is responsible for overseeing that every step of the process stays within the specified quality parameters. The guidelines are the standards for each product and for each step of the process necessary to produce the highest quality feed at the optimum capacity. The QA department – and not the production manager – has the final word on what products are cleared for sale or dispatching to the client.

Proportioning cost center
In the proportioning cost center, all the ingredients in the formula are weighed to constitute a formula mix, which is the product. To ensure that the process is properly done, the operator must register the sequence of ingredients weighed and make sure that their weights correspond to the formula.
In this cost center, the operator is also responsible for transferring raw materials from the bulk storage section (warehouse or silos) to the proportioning bins. Also during this step, the operator is responsible for checking that all magnets and scalpers are cleaned. The operator is also responsible for making sure that the ingredients are placed in the right bin to avoid cross contamination, checking that the scales are functioning properly and, if not, reporting it to the maintenance personnel.
Similarly, the operator must ensure that the dosing bins are maintained and kept cleaned, and that no material hang-ups are left spoiling in the dead spots of the bin. At the same time, QA works together with the operator to monitor the process by collecting samples of the ingredients that will be later analyzed to determine if cross contamination has occurred. Good record keeping is critical at every step of the process.
The QA person must record the time, date, formula identification number, and batch number. The operator, at the same time, labels all the samples to the corresponding formula identification number, and batch number. This information can be later correlated to the rest of the process and be used as a tool to determine if anything was improperly done or was out of standards.
The most common mistake in the proportioning cost center is the inclusion of the wrong raw material as the result of dosing from the wrong bin, or simply because the operator filled the bin with the wrong ingredient. A careful analysis, with proper record keeping, can prevent a finished product, outside the standards, reaching the end user, the shrimp farm. In most cases, these out-of-standard products should be prevented from leaving the plant, or be recalled in case the plant has already dispatched them to the farm.
However, the latter should never be allowed. It is critical to have the product cleared for sale by QA prior to dispatching. The best way to accomplish the latter is to have enough finished product capacity to store the feed for at least a week to give QA plenty of time to do all the respective analyses. Thus, the production manager should schedule dispatching finished products based on a first-in first out (FIFO) schedule.
Therefore, once QA has cleared a finished product, it should be allowed for delivery based on production date, including batch and formula number. Recalled product that is still considered fresh based on the quality standards can be reutilized in the formulas.
The first step for including a finished product in the formula is to know its nutritional parameters (crude protein, fat, ash, etc.), its cost (including reprocessing costs) and its overall quality. Once the finished product is clear for use as an ingredient, it should be assigned to a reprocess bin and included in the formulation using the same strict guidelines set forth by the company.
Since this already is a processed ingredient, its inclusion level should not exceed more than 2 percent to avoid affecting other physical parameters such as water stability. The inclusion of these-out-of-standard finished products or products that did not clear QA/QC for reasons other than sanitary (positive results on mycotoxins or pathogenic microorganisms) is permissible, and they can therefore be re-used by the aquafeed mill.

Grinding cost center
Once the pre-blended mix reaches the grinding cost center, the process is now owned by the mill operator. This cost center is the most critical step of the process, for any deviation from the target particle size can drastically affect the process downstream, and this can also affect the nutritional and physical properties of the aquafeed. In this cost center, the operator is accountable for the proper maintenance and functioning of the grinding equipment, and also to ensure that the blend being ground is within the standards.
Fig. 1 shows a record for data collection in the grinding cost center, set up to have the target particle size and the upper (UL) and lower limit (LL). The standards for the particle size limits should be based on previous optimization runs with similar ingredients. Once the processing parameters and standards are established, the operator should be able to obtain the same quality product with small modifications in the processing parameters due to wear of the screens and hammers, as well as changes in the physical properties of some ingredients in the formula.
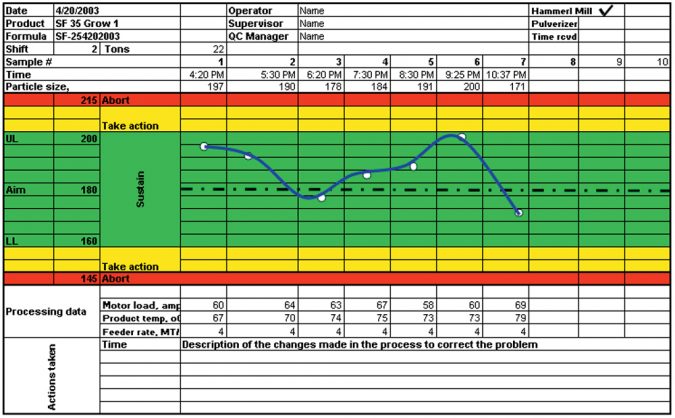
In the grinding process, it is critical to collect data at least every hour. This can be done by setting up sieve analysis in the grinding room that will give the operator the tool to monitor the process in real time. The operator, upon completing the sieve analysis, should save a sample in a bag labeled with the date, time, and formula identification code, and submit the samples to the QA department. The record in Fig. 1 is typical for a process running under normal conditions, in which the product attributes stay within the green optimum range.
In contrast, in the record shown in Fig.2, the process is out of the specified green range four hours into the run. It is obvious from the data that something was wrong and caused the particle size to be above the UL. According to the data shown, the motor load dropped to 40 amps from a normal rate of 63-70 amps load. This is indicative of a broken screen, which the operator was able to detect on time thanks to the in-line process control with the sieve analysis. Once the grinding was resumed with the new screen, the operator could determine that the particle size was back in the green range.
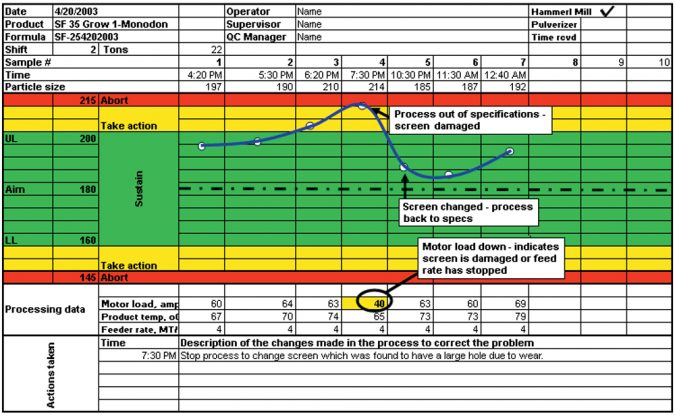
The in-line quality control with proper data collection can also be an important tool for preventive maintenance. The data can tell the operator the approximate run hours or tons produced required to wear out the parts. This information is useful to plan and schedule ahead of time any screens or hammers changes (rotate the hammers or screens) necessary. The operator can take preventive measures to avoid surprises in the process that can increase the down time and reduce the overall efficiency of the grinding cost center.
Perspectives
Overall process quality control is a commitment that needs to be embraced by the whole organization, from the its president to the operators, and the personnel should be properly trained and have clear accountabilities in their respective cost centers. It is also very important to have the necessary testing equipment, defined product standards and process parameters, and all the necessary forms to record process performance. And the quality control department should also perform periodic compliance inspections to ascertain that the company’s quality standards are being continuously met.
Author
-

Eugenio Bortone, Ph.D., PAS, Dpl. ACAN
Sapientia Technology
Frisco, Texas USA[109,111,99,46,108,105,97,109,103,64,101,110,111,116,114,111,98,114,100]
Tagged With
Related Posts

Aquafeeds
Twin screw technology for better extruded feeds
Extrusion processing can be a better alternative to produce high quality-low- polluting shrimp feeds. Extruded pellets with high water stability can be produced by controlling the Specific Mechanical Energy (SME) and without the inclusion of artificial binders. New extruders are equipped with state-of-the art SME controls that permit the adjustment of the SME to the desired range, to produce soft very hydrostable pellets.

Aquafeeds
A look at the SME controlled extrusion process
A study was conducted using a Twin-Screw Extruder equipped with Specific Mechanical Energy (SME) and Density Control valves, to determine the effect of SME on the water stability of shrimp feeds. Further research is needed to evaluate the performance.

Aquafeeds
Shrimp feeds depend on proper ingredient grinding
The proper feed particle sizes of aquafeed ingredients are established by the nutritional requirements of the species targeted and by the feed manufacturing process used. For proper ingredient particle sizes, various characteristics are very important, including particle size distribution and pellet density.

Aquafeeds
Alternative feed ingredient universe to convene at F3 meeting
What started out as a simple yet ambitious contest to drive innovation in the aquafeed sector has evolved into a fully global competition – and collaboration – amongst ingredient suppliers and feed manufacturers.

